Fasting Part 1 gave some background information about what happens during fasting and how to prepare for a fast. So which fasting method should you choose? We dive deep into the research below and give you the specific methods appropriate for goals including fat loss, longevity, and others.
Below you can find:
- Fasting Goals – What Does the Research Say?
- Enhancing Longevity and Healthspan
- Making New Stem Cells
- Enhancing Autophagy
- Treating Cancer
- Summary: The Takeaways
This is Part 2 of the Ketosource series on fasting.
If you haven’t read it yet, Fasting Part 1 gives some background information on fasting. Part 1 describes what happens during fasting and how to prepare for a fast. Take a look at Part 1 to get some foundation before continuing with Fasting Part 2 here.
Below you will learn which fasting method is best for each fasting goal.
Check out the Summary Takeaways section at the end to see how fasting fits your specific goals. Then come back and grab all the details and references to go over with your doctor. That way you’ll be fully prepared and get the most from your fast.
Fasting Goals – What Does the Research Say?
As you’ll see below, guidelines for some of the major fasting goals are clearly defined by research on humans.
But this is not true for all the fasting goals. Some of these goals have only been demonstrated in animal models so far.
For some of these goals, it is still possible to create reasonable guidelines based on animal models. As discussed above, markers of the Fasted State help to compare the fasting timelines for humans and other animals.
Some of the major fasting goals are explained below. For each goal, the relevant fasting methods are outlined in detail. These details are also summarized in the Takeaways section at the end.
Enhancing Longevity and Healthspan
Increased longevity is often considered the Holy Grail of benefits from lifestyle interventions.
Many expect this benefit from fasting. After all, animals like nematodes and rats see longevity benefits from calorie restriction and fasting.
Nematodes and rats are separated by over 900 million years of evolution! There must be something about this type of restriction that is inherently good for longevity.
But even if fasting does increase human longevity, it would be difficult to measure. Longevity studies in humans are not practical due to the necessary time and resources.
If lifespan is the “years in your life”, then healthspan is the “life in your years.” Healthspan is a way to refer to healthy aging, i.e. how many years you can live without the burden of disease.
Markers of healthspan are much easier to measure in humans than lifespan.
Alternate-Day Fasting
Increased longevity was found in fasting rats as early as 1946. These rats fasted at least one day a week. Rats who fasted every one in three days had the best combination of healthspan benefits and increased longevity.
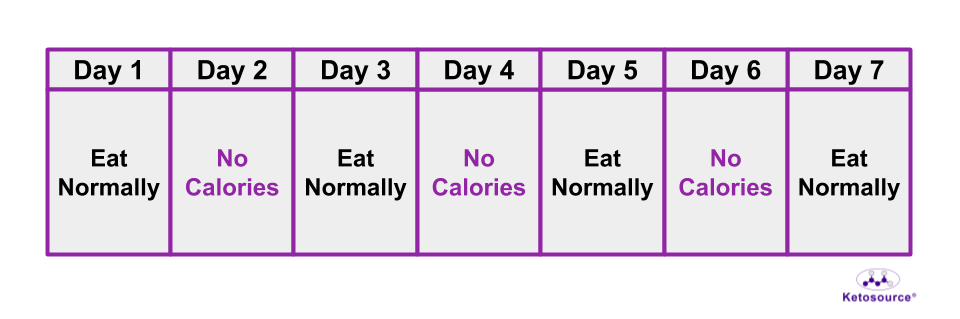
This fasting method is most similar to alternate-day fasting in humans. You can see an illustration of alternate-day fasting in Figure 10 below.
The 1946 study on fasting rats looked at breast tumors as a marker of healthspan. These tumors were assessed in female rats in the control and fasting groups. The female rats who fasted every one in three days developed smaller tumors than the control group. These tumors also appeared later in life than tumors in the control group.
Figure 10. One week of the alternate-day fasting method.
The Fasting Mimicking Diet (FMD) and Prolonged Fasting
The Fasting Mimicking Diet (FMD) improves the healthspan of mice. Like the rat study above, this includes a decrease in the number of mice with tumors. It’s important to note here that the FMD for mice is different than the FMD for humans.
(NOTE: Fasting Mimicking Diet, FMD, and Prolon are registered trademarks of L-Nutra)
For mice, this version of the FMD is four days long and begins with ~50% of normal calories on Day One. Then the mice eat ~10% of normal calories on Days 2-4.
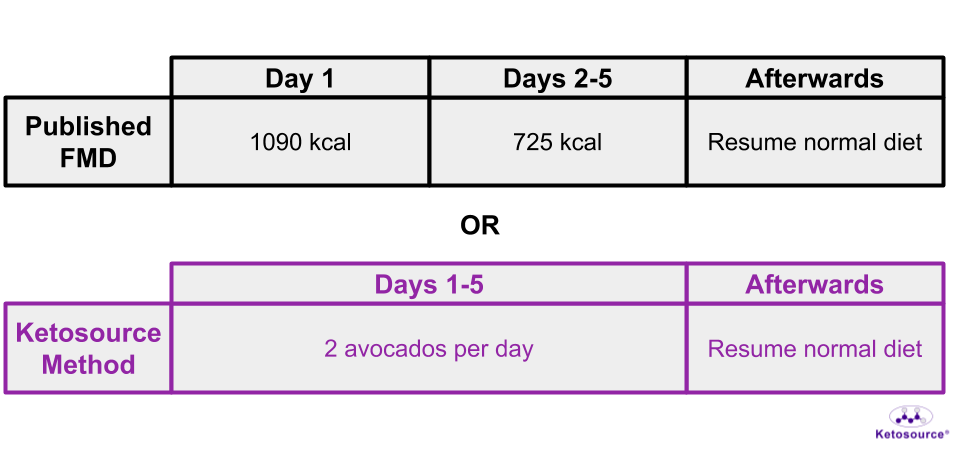
For humans, this version of the FMD is five days long with ~1090 calories on Day One and ~725 calories on Days 2-5.
If you follow a 2000 calorie/day diet, this means you’d eat ~53% of your normal calories on Day One and ~36% of your normal calories on Days 2-5.
You can see that the mouse FMD has a deeper Fasted State than the human FMD based on the % drop in calories.
As you saw in the fasting timeline graphs, ketone bodies serve as a marker of the Fasted State. Levels of ketone bodies increase ~10-fold in mice at the end of their FMD cycle. In humans, this increase is only 3.7-fold.
To see ketone body changes like the mice, you would need to fast for at least two days.
IGF1 serves as marker of the Fed State and goes down while fasting. IGF1 levels decrease by ~45% in mice by the end of their FMD cycle. In humans, IGF1 levels decrease by only 24%.
To get IGF1 levels to drop by 45% like the mice, you would need to fast for at least 2-3 days. There is a large variation in how people’s IGF1 levels drop during a fast though, so this may take as long as 5 days.
Another important difference in the mouse and human FMDs is how often they are performed. In this study, mice repeated the FMD every two weeks once they hit middle age. The humans in this study followed the FMD once a month for only three months.
This is not to say the 5-day FMD is useless for humans. In humans, three months of the 5-day FMD improves several biomarkers which can predict disease. If you have biomarkers trending in the wrong direction, the FMD can be useful to get you back on track.
But to get some of the same healthspan benefits that mice get from the FMD, three days of fasting would be a good place to start.
Using a 3-day fast once per month for three consecutive months may be even more beneficial than the current 5-day FMD method. This is based on the changes in blood glucose, ketones, and IGF1 levels. But there is no study that has tested this yet.
Alternatively, you could also modify the FMD to focus your calories on high-fat and high-fiber foods. The human 5-day FMD method includes carbohydrates and protein which take you out of the Fasted State.
An avocado-based FMD is described on The Quantified Body podcast which you can find here. This will be referred to as the Ketosource Method from now on. Some results from one cycle of the Ketosource Method show an even deeper Fasted State than the mouse FMD. You can see a breakdown of the published FMD and the Ketource Method in Figure 11 below.
Interestingly, IGF1 levels dropped by 44% in the Ketosource Method. This suggests that one cycle of the Ketosource Method is at least equivalent to one cycle of the mouse FMD.
To get even more benefits, you could do two or more cycles of the Ketosource Method every month.
Figure 11. Different approaches to the 5-day FMD.
Time-Restricted Feeding (TRF)
The FMD was developed to mimic prolonged fasting. But it is not the first diet designed to do this. Over a century ago, the ketogenic diet was developed as a fasting replacement for the treatment of epilepsy.
Recent mouse studies on the ketogenic diet showed improved mid-life mortality and median lifespan. One key difference between these two studies is how they dealt with food intake.
Roberts, et al. controlled the calorie intake of mice, which means the researchers likely fed the mice at the same time every day. In this way, the median lifespan increase for these mice could have come from TRF.
In support of this, mice eating one meal a day have increased longevity and healthspan. This seems to be independent of what they eat or how much they eat.
As mentioned already, the fasting timelines for mice and humans are very different. There is no great way to compare these benefits for mice with potential TRF benefits for humans.
But there are some modest benefits for people following TRF.
For example, most people eat within a 15-hour window that extends into the night. Eating between 6 AM and 7 PM could prevent overeating and lead to weight loss.
An 11-hour eating window (13-hour overnight fast) is also associated with less breast cancer recurrence.
An 8-hour eating window shows some benefit for obese people and even for athletes.
Those with prediabetes or at risk for type 2 diabetes can see benefits from eating within 6 hours and 9 hours, respectively. In these cases, the eating window should begin in the morning (8-9 AM).
Making New Stem Cells
One of the most overlooked benefits of fasting is its effect on stem cells.
When you get an injury, stem cells replace the dead cells and regenerate the damaged area. But it is well known that this ability of stem cells decreases with age.
Hematopoietic stem cells, or stem cells of the blood system, illustrate this well.
The number of hematopoietic stem cells in mice increases as they age. But hematopoietic stem cells from aged mice are more defective than the same cells from young mice.
Prolonged Fasting
Water-only fasting for three days in mice improves the function of their hematopoietic stem cells.
This length of fasting causes the mice to create more stem cells. But unlike aging, in this case the new stem cells are more functional than the old stem cells.
This was evident from the ratio of blood cell types which looked more like young mice after fasting. In this way, fasting “rejuvenated” the blood systems of these mice.
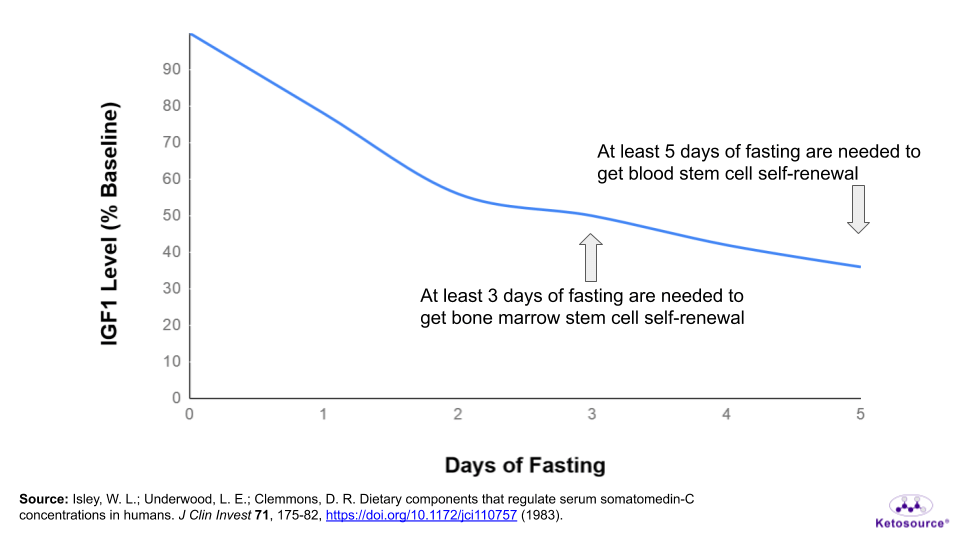
These benefits are believed to come from a drop in IGF1 levels in the mice. At the end of a 3-day fast, IGF1 levels in mice drop by 70%.
You would need to fast for at least 5 days to get a similar drop in IGF1 levels. As stated earlier, people’s IGF1 levels drop at different rates during fasting. It could take as long as 9 days of fasting to see this drop in IGF1.
The Fasting Mimicking Diet (FMD)
The FMD also provides stem cell benefits to mice. This includes benefits to brain stem cells and stem cells found in the bone marrow.
Interestingly, certain organs shrink (kidney, heart, liver) in mice while they follow the FMD. These organs then return to their normal weights or heavier after the mice eat again for a week. There are also signs of organ and blood system rejuvenation following this re-feeding phase.
This highlights the importance of the re-feeding phase. Without entering back into this Fed State after the Fasted State, the mice would not be able to use their benefits from their Fasted State.
As discussed in the “Enhancing Longevity and Healthspan” section, the mouse FMD has a deeper Fasted State than the human 5-day FMD. But you can mimic the mouse FMD with water-only fasting for 3-5 days or by using the Ketosource Method as an FMD.
Figure 12. Where stem cell benefits fall on the fasting timeline.
Enhancing Autophagy
Autophagy is the process whereby old or damaged cellular machinery gets recycled.
Autophagy is integral to many cellular functions, and a background level of autophagy is found in practically all cells. This means autophagy benefits come from ways to “enhance” autophagy, which already takes place to some degree.
Certain natural compounds which promote longevity in multiple species do so via autophagy. Mouse studies have also found that coffee and exercise enhance autophagy. Unfortunately, no studies have confirmed these findings in humans yet.
However, water-only fasting can enhance autophagy in both mice and humans.
Research suggests the timelines are different for each species. And that the timelines are also unique for different cells in the body.
Autophagy is difficult to address as a fasting goal. One reason for this is how complex autophagy is to measure.
As explained below, insulin levels are the best way to compare the autophagy timelines for mice and humans right now. There is a lot of interest in tracking autophagy in humans, so insulin levels could be one way to track this.
Prolonged Fasting
In mice, autophagy is enhanced in muscle cells after one day of fasting, and this is shut off by the hormone insulin. This suggests that insulin levels can be used to compare the animal and human timelines for autophagy.
After one day of fasting, levels of insulin in mice have dropped by 95%.
You would need to fast for at least 2 days to get this drop in insulin. However, older human studies suggest that a 95% drop in insulin does not happen during fasting. Of course, the starting point for insulin could also matter here.
For mice, this 95% drop in insulin could be a plateau. This means it could be the lowest insulin level possible for mice. In this case, the human equivalent would likely be 5 days of fasting.
In support of this, there are mixed signals for autophagy in human muscle cells after 3 days of fasting.
This could be because insulin has not dropped enough yet to enhance autophagy. Or it could be due to the difficulty in measuring autophagy and interpreting data for the protein p62.
Autophagy usually degrades p62. The muscle cells showed some signs of autophagy in this study, but they also had higher levels of p62.
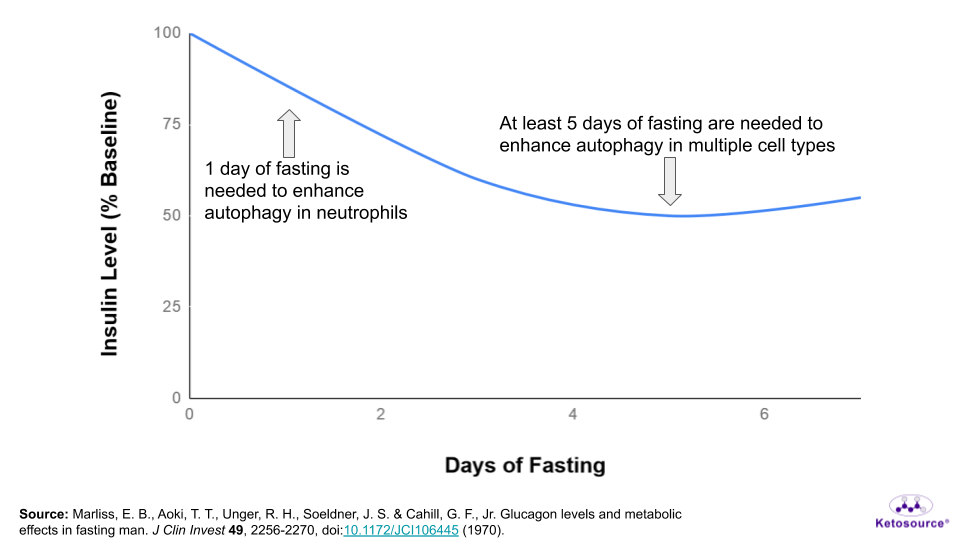
A study on white blood cells found that autophagy might be enhanced in many types of human white blood cells within 4 days of fasting. But detailed testing was only carried out after one day of fasting. This detailed testing revealed that autophagy is enhanced in just one type of white blood cell (the neutrophil) in humans after one day of fasting.
In mice, autophagy is enhanced in all types of white blood cells within two days of fasting. Could these autophagy benefits have started earlier in the fast? The researchers did not check. So for now, there is no great way to compare autophagy timelines yet for white blood cells.
Taken together, insulin is the only trackable biomarker to use when comparing the human and mouse autophagy timelines.
So for now, these studies suggest that you would need to fast for at least one day to enhance autophagy in one cell type (the neutrophil).
But based on the muscle studies and insulin levels during fasting, most likely at least five days of fasting are necessary to get autophagy benefits in multiple cell types. This is shown in the graph below.
Figure 13. Where autophagy benefits fall on the fasting timeline.
The Fasting Mimicking Diet (FMD)
There are signs of muscle autophagy benefits in mice following the FMD.
Mice following the FMD have decreased expression of the protein p62 in muscle. As stated earlier, p62 is degraded by autophagy.
However, this result was also seen in mice who had followed cycles of the FMD consistently for a long time. And the level of p62 in these mice on the FMD was similar to that of young mice not on the FMD.
This suggests that mice who regularly follow the FMD are more able to undergo muscle autophagy than normal mice of the same age.
Based on the role of p62 in tumor formation, this might also help explain why these mice had fewer tumors when they died.
Autophagy markers were not measured in humans in the study. And like the mouse study on muscle autophagy, the insulin levels for mice drop by ~90% during the FMD.
As stated earlier, this level of insulin is seen in humans after 5 days of fasting. This suggests that regularly fasting for 5 days could enhance your long-term autophagy benefits. In other words, it could ensure you maintain the ability to undergo autophagy as you age.
Treating Cancer
This Is Not a Recommendation
The use of fasting in cancer treatment is very controversial. This section is not intended to promote the use of fasting as an adjuvant to, or as a replacement for cancer treatment.
Nor is it intended to offer guidelines for treating cancer. It is intended only to serve as reference material which you can go over with your physician.
As mentioned above, the Fasted State comes with lower levels of the pro-growth hormones IGF1 and insulin. The Fasted State also comes with lower blood glucose levels and higher blood ketone body levels.
Insulin and IGF1 are both implicated in the development of cancer and in its progression. Inhibiting the pro-growth effects of these hormones is a major focus in cancer drug development.
Similarly, glucose metabolism is another target for cancer therapy. An estimated >90% of cancers show signs of enhanced glucose metabolism.
Studies suggest that cancer arises due to problems with cellular metabolism. And that these problems make cancer cells rely on glucose.
During fasting, normal cells can deal with a shift from glucose metabolism to ketone body metabolism. In fact, nearly 60% of brain metabolism shifts to using ketone bodies during long fasts.
Glucose levels as low as 0.5 mM (9 mg/dL) have been recorded during fasting without symptoms of hypoglycemia. This is only ~10% of a normal blood glucose level. This low blood glucose level would be fatal without the high levels of ketone bodies that come from fasting.
As stated above, lowering glucose and pro-growth hormones while increasing ketone bodies is a key feature of the Fasted State. This is believed to selectively harm cancer cells.
Prolonged Fasting
Mice with 70-80% lower IGF1 levels are more protected against the side effects of chemotherapy than normal mice. These mice also have better survival against melanoma. This is the IGF1 level reached by normal mice after fasting for three days.
Mice who fast for three days are also protected against the side effects of chemotherapy. Together, these studies suggest that a ~70% drop in IGF1 levels is beneficial for chemotherapy side effects and cancer survival in mice.
As mentioned earlier, humans need to fast for at least 5 days to see this drop in IGF1 levels.
Have Cancer Patients Fasted for Five Days?
There are at least three studies on prolonged fasting in cancer patients. In each of these studies, the fasting period was right before and/or right after chemotherapy. Two of the studies were on the same group of patients.
None of the studies focused on fasting as a treatment for cancer. But each of them suggested that fasting can be safe for cancer patients undergoing chemotherapy.
In one of these studies, ten patients with various cancers fasted for 48-140 hours before and/or 5-56 hours after chemotherapy. On average, fatigue and weakness from the chemotherapy were better when the patients were fasting. None of the chemotherapy side effects became worse.
For some of the patients of this study, the effectiveness of their chemotherapy could be measured. In these cases, there were no signs that fasting prevented the effectiveness of chemotherapy against their cancers.
In the second study, 20 patients with various cancers fasted for 1-3 days surrounding chemotherapy. This fasting period allowed for up to 200 calories per day. The changes in glucose, ketones, and pro-growth hormones varied amongst the patients.
Like the mouse study mentioned above, there were benefits for white blood cells in patients who fasted for 2-3 days. Specifically, their white blood cells were protected against DNA damage from the chemotherapy.
The third study looked at the same group of patients as the second study. This third study also found that white blood cells were more protected in patients who fasted for three days compared to patients who fasted for one day.
Together, these three studies suggest that fasting can be safe for cancer patients and it can help with the side effects of chemotherapy. These studies also suggest that fasting for 2-3 days can help protect the immune system from chemotherapy side effects.
But it’s important to remember that these fasts were carried out under medical supervision. Malnutrition and muscle wasting are still some of the major concerns for the use of fasting in cancer treatment. More research is needed before a specific fasting method is proven safe and effective for cancer patients.
Summary: The Takeaways
The best fasting method for you is based on your specific goal(s), lifestyle, and any medications you currently take.
Takeaways: How to Achieve Your Fasting Goals
-
Weight Loss: Use alternate-day fasting to lose weight. This method has a high compliance rate and keeps you at a big calorie deficit for the week.
-
Enhancing Longevity and Healthspan: Use 3 monthly cycles of the Ketosource Method (found here) to get disease markers under control. Use a 5-day fast in place of the FMD to add autophagy and stem cell benefits. Repeat as necessary if your blood markers start trending in the wrong direction.
-
Enhancing Autophagy: A 1-day fast will enhance autophagy in one cell type (the neutrophil). Use a 5-day fast to enhance autophagy for multiple cell types.
-
Stem Cell Self-Renewal: Use a 5-day fast to promote stem cell self-renewal. Depending on how IGF1 levels drop for you, this could require up to nine days of fasting.
-
Treating Cancer: More research is needed to clarify the safety and efficacy of fasting in cancer patients. It is too early to set any takeaways for the role of fasting in cancer treatment.
-
Treating Type 2 Diabetes: Use modified fasting so you can take your medications safely. Use more than two low-calorie days per week to lose weight more quickly. Add an early 6-9 hour TRF on top of this fasting method to get more benefit.
References
{5748374:JE5J8BEP},{5748374:83UM7QL8};{5748374:ES9NPU2S},{5748374:MF4ZZAX3};{5748374:TYQH7SPE};{5748374:ES9NPU2S};{5748374:ES9NPU2S};{5748374:3HHU6VZN};{5748374:ES9NPU2S};{5748374:3HHU6VZN};{5748374:3HHU6VZN};{5748374:3HHU6VZN};{5748374:3HHU6VZN};{5748374:9R8B7N39};{5748374:3HHU6VZN};{5748374:P2Z6B7GC};{5748374:E6IMUMCA};{5748374:3HHU6VZN};{5748374:SQU9ZGIQ};{5748374:SQU9ZGIQ},{5748374:3HHU6VZN},{5748374:WX6XVYQC},{5748374:P2Z6B7GC};{5748374:3HHU6VZN};{5748374:3HHU6VZN},{5748374:WZLT2AEX};{5748374:WZLT2AEX};{5748374:3HHU6VZN};{5748374:3HHU6VZN};{5748374:VMKSQVPV};{5748374:9N4I95G9};{5748374:3YQ4G2FK};{5748374:3YQ4G2FK};{5748374:HZ5GBZQF};{5748374:HZ5GBZQF};{5748374:8J8FRJPG};{5748374:CXS8BFWR};{5748374:S6L3EE64};{5748374:CUMUP9TI};{5748374:XIPWH7MH};{5748374:C6HDVJ7S};{5748374:4NRKPFQR};{5748374:J7ZVR9HH};{5748374:4NB77YNV};{5748374:6NCTSX67};{5748374:XL67M6CI};{5748374:3WVQ9RWW};{5748374:XL67M6CI};{5748374:3WVQ9RWW};{5748374:3WVQ9RWW};{5748374:3WVQ9RWW};{5748374:338PM6JJ};{5748374:P2Z6B7GC};{5748374:E6IMUMCA};{5748374:3HHU6VZN};{5748374:3HHU6VZN};{5748374:3HHU6VZN},{5748374:P2Z6B7GC};{5748374:WZLT2AEX};{5748374:8FYI8Q6H};{5748374:R2BJ3B97},{5748374:CE2PQYYN};{5748374:ZGACAGRI};{5748374:L5K2CMYB};{5748374:4XGWAUUR};{5748374:P4DNECM5};{5748374:TWZWWXS5};{5748374:9R8B7N39};{5748374:TWZWWXS5};{5748374:4TLPENTP};{5748374:3XRK86DQ};{5748374:AAWHJF5X};{5748374:I2Y8YCZK};{5748374:3XRK86DQ};{5748374:XTBGIK8S};{5748374:XTBGIK8S};{5748374:XTBGIK8S};{5748374:3XRK86DQ},{5748374:P4DNECM5};{5748374:TWZWWXS5},{5748374:4TLPENTP};{5748374:3HHU6VZN};{5748374:3HHU6VZN};{5748374:I2Y8YCZK};{5748374:3HHU6VZN};{5748374:AFC6LMAY};{5748374:3HHU6VZN};{5748374:3HHU6VZN};{5748374:TWZWWXS5},{5748374:P4DNECM5};{5748374:3HHU6VZN};{5748374:4TLPENTP};{5748374:GJB9K3L2};{5748374:P2Z6B7GC};{5748374:WX6XVYQC};{5748374:WX6XVYQC};{5748374:MDJEWPB4};{5748374:UMACU7KS};{5748374:HRIQWFPR};{5748374:PWKABTDZ};{5748374:UMACU7KS};{5748374:ZA4XJNFE};{5748374:QWP452KJ};{5748374:9R8B7N39};{5748374:WX6XVYQC},{5748374:E6IMUMCA};{5748374:UMACU7KS};{5748374:338PM6JJ};{5748374:3WVQ9RWW};{5748374:E6IMUMCA},{5748374:P2Z6B7GC};{5748374:NCTGYSIU},{5748374:ZZT2RDW2},{5748374:3WVQ9RWW};{5748374:NCTGYSIU};{5748374:NCTGYSIU};{5748374:ZZT2RDW2};{5748374:3WVQ9RWW};{5748374:ZZT2RDW2};{5748374:3WVQ9RWW};{5748374:ZZT2RDW2},{5748374:3WVQ9RWW},{5748374:NCTGYSIU};{5748374:GJB9K3L2}
3d-printed-materials-and-systems
asc
0
8655
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3A%22zotpress-0eab6d7d693aeca30fecfbb94188c443%22%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22C6HDVJ7S%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Moro%20et%20al.%22%2C%22parsedDate%22%3A%222016%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EMoro%20T%2C%20Tinsley%20G%2C%20Bianco%20A%2C%20et%20al%20%282016%29%20Effects%20of%20eight%20weeks%20of%20time-restricted%20feeding%20%2816%5C%2F8%29%20on%20basal%20metabolism%2C%20maximal%20strength%2C%20body%20composition%2C%20inflammation%2C%20and%20cardiovascular%20risk%20factors%20in%20resistance-trained%20males.%20J%20Transl%20Med%2014%3A290.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1186%5C%2Fs12967-016-1044-0%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1186%5C%2Fs12967-016-1044-0%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Effects%20of%20eight%20weeks%20of%20time-restricted%20feeding%20%2816%5C%2F8%29%20on%20basal%20metabolism%2C%20maximal%20strength%2C%20body%20composition%2C%20inflammation%2C%20and%20cardiovascular%20risk%20factors%20in%20resistance-trained%20males%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tatiana%22%2C%22lastName%22%3A%22Moro%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Grant%22%2C%22lastName%22%3A%22Tinsley%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Antonino%22%2C%22lastName%22%3A%22Bianco%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Giuseppe%22%2C%22lastName%22%3A%22Marcolin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Quirico%20Francesco%22%2C%22lastName%22%3A%22Pacelli%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Giuseppe%22%2C%22lastName%22%3A%22Battaglia%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Antonio%22%2C%22lastName%22%3A%22Palma%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Paulo%22%2C%22lastName%22%3A%22Gentil%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Marco%22%2C%22lastName%22%3A%22Neri%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Antonio%22%2C%22lastName%22%3A%22Paoli%22%7D%5D%2C%22abstractNote%22%3A%22BACKGROUND%3A%20Intermittent%20fasting%20%28IF%29%20is%20an%20increasingly%20popular%20dietary%20approach%20used%20for%20weight%20loss%20and%20overall%20health.%20While%20there%20is%20an%20increasing%20body%20of%20evidence%20demonstrating%20beneficial%20effects%20of%20IF%20on%20blood%20lipids%20and%20other%20health%20outcomes%20in%20the%20overweight%20and%20obese%2C%20limited%20data%20are%20available%20about%20the%20effect%20of%20IF%20in%20athletes.%20Thus%2C%20the%20present%20study%20sought%20to%20investigate%20the%20effects%20of%20a%20modified%20IF%20protocol%20%28i.e.%20time-restricted%20feeding%29%20during%20resistance%20training%20in%20healthy%20resistance-trained%20males.%5CnMETHODS%3A%20Thirty-four%20resistance-trained%20males%20were%20randomly%20assigned%20to%20time-restricted%20feeding%20%28TRF%29%20or%20normal%20diet%20group%20%28ND%29.%20TRF%20subjects%20consumed%20100%5Cu00a0%25%20of%20their%20energy%20needs%20in%20an%208-h%20period%20of%20time%20each%20day%2C%20with%20their%20caloric%20intake%20divided%20into%20three%20meals%20consumed%20at%201%5Cu00a0p.m.%2C%204%5Cu00a0p.m.%2C%20and%208%5Cu00a0p.m.%20The%20remaining%2016%5Cu00a0h%20per%2024-h%20period%20made%20up%20the%20fasting%20period.%20Subjects%20in%20the%20ND%20group%20consumed%20100%5Cu00a0%25%20of%20their%20energy%20needs%20divided%20into%20three%20meals%20consumed%20at%208%5Cu00a0a.m.%2C%201%5Cu00a0p.m.%2C%20and%208%5Cu00a0p.m.%20Groups%20were%20matched%20for%20kilocalories%20consumed%20and%20macronutrient%20distribution%20%28TRF%202826%5Cu00a0%5Cu00b1%5Cu00a0412.3%5Cu00a0kcal%5C%2Fday%2C%20carbohydrates%2053.2%5Cu00a0%5Cu00b1%5Cu00a01.4%5Cu00a0%25%2C%20fat%2024.7%5Cu00a0%5Cu00b1%5Cu00a03.1%5Cu00a0%25%2C%20protein%2022.1%5Cu00a0%5Cu00b1%5Cu00a02.6%5Cu00a0%25%2C%20ND%203007%5Cu00a0%5Cu00b1%5Cu00a0444.7%5Cu00a0kcal%5C%2Fday%2C%20carbohydrates%2054.7%5Cu00a0%5Cu00b1%5Cu00a02.2%5Cu00a0%25%2C%20fat%2023.9%5Cu00a0%5Cu00b1%5Cu00a03.5%5Cu00a0%25%2C%20protein%2021.4%5Cu00a0%5Cu00b1%5Cu00a01.8%29.%20Subjects%20were%20tested%20before%20and%20after%208%5Cu00a0weeks%20of%20the%20assigned%20diet%20and%20standardized%20resistance%20training%20program.%20Fat%20mass%20and%20fat-free%20mass%20were%20assessed%20by%20dual-energy%20x-ray%20absorptiometry%20and%20muscle%20area%20of%20the%20thigh%20and%20arm%20were%20measured%20using%20an%20anthropometric%20system.%20Total%20and%20free%20testosterone%2C%20insulin-like%20growth%20factor%201%2C%20blood%20glucose%2C%20insulin%2C%20adiponectin%2C%20leptin%2C%20triiodothyronine%2C%20thyroid%20stimulating%20hormone%2C%20interleukin-6%2C%20interleukin-1%5Cu03b2%2C%20tumor%20necrosis%20factor%20%5Cu03b1%2C%20total%20cholesterol%2C%20high-density%20lipoprotein%20cholesterol%2C%20low-density%20lipoprotein%20cholesterol%2C%20and%20triglycerides%20were%20measured.%20Bench%20press%20and%20leg%20press%20maximal%20strength%2C%20resting%20energy%20expenditure%2C%20and%20respiratory%20ratio%20were%20also%20tested.%5CnRESULTS%3A%20After%208%5Cu00a0weeks%2C%20the%202%20Way%20ANOVA%20%28Time%5Cu00a0%2A%5Cu00a0Diet%20interaction%29%20showed%20a%20decrease%20in%20fat%20mass%20in%20TRF%20compared%20to%20ND%20%28p%5Cu00a0%3D%5Cu00a00.0448%29%2C%20while%20fat-free%20mass%2C%20muscle%20area%20of%20the%20arm%20and%20thigh%2C%20and%20maximal%20strength%20were%20maintained%20in%20both%20groups.%20Testosterone%20and%20insulin-like%20growth%20factor%201%20decreased%20significantly%20in%20TRF%2C%20with%20no%20changes%20in%20ND%20%28p%5Cu00a0%3D%5Cu00a00.0476%3B%20p%5Cu00a0%3D%5Cu00a00.0397%29.%20Adiponectin%20increased%20%28p%5Cu00a0%3D%5Cu00a00.0000%29%20in%20TRF%20while%20total%20leptin%20decreased%20%28p%5Cu00a0%3D%5Cu00a00.0001%29%2C%20although%20not%20when%20adjusted%20for%20fat%20mass.%20Triiodothyronine%20decreased%20in%20TRF%2C%20but%20no%20significant%20changes%20were%20detected%20in%20thyroid-stimulating%20hormone%2C%20total%20cholesterol%2C%20high-density%20lipoprotein%2C%20low-density%20lipoprotein%2C%20or%20triglycerides.%20Resting%20energy%20expenditure%20was%20unchanged%2C%20but%20a%20significant%20decrease%20in%20respiratory%20ratio%20was%20observed%20in%20the%20TRF%20group.%5CnCONCLUSIONS%3A%20Our%20results%20suggest%20that%20an%20intermittent%20fasting%20program%20in%20which%20all%20calories%20are%20consumed%20in%20an%208-h%20window%20each%20day%2C%20in%20conjunction%20with%20resistance%20training%2C%20could%20improve%20some%20health-related%20biomarkers%2C%20decrease%20fat%20mass%2C%20and%20maintain%20muscle%20mass%20in%20resistance-trained%20males.%22%2C%22date%22%3A%2210%2013%2C%202016%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1186%5C%2Fs12967-016-1044-0%22%2C%22ISSN%22%3A%221479-5876%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22UIM8YXRX%22%2C%22JJ3BUIE2%22%2C%226BPRM5LB%22%2C%227RQSZ6D8%22%5D%2C%22dateModified%22%3A%222019-08-15T03%3A45%3A54Z%22%7D%7D%2C%7B%22key%22%3A%22WZLT2AEX%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Blenkinsopp%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EBlenkinsopp%20D%20The%205-day%20fast%20mimicking%20diet%20%28self%20experiment%29.%20The%20Quantified%20Body%20Podcast.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22podcast%22%2C%22title%22%3A%22The%205-day%20fast%20mimicking%20diet%20%28self%20experiment%29.%20The%20Quantified%20Body%20Podcast.%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22podcaster%22%2C%22firstName%22%3A%22Damien%22%2C%22lastName%22%3A%22Blenkinsopp%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22episodeNumber%22%3A%2231%22%2C%22audioFileType%22%3A%22mp3%22%2C%22runningTime%22%3A%22%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fthequantifiedbody.net%5C%2Ffast-mimicking-diet%5C%2F%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%22JZKZ7F4X%22%5D%2C%22dateModified%22%3A%222019-07-18T02%3A47%3A28Z%22%7D%7D%2C%7B%22key%22%3A%22HZ5GBZQF%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Woo%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EWoo%20G%20mTor%20Signaling%20%26amp%3B%20Cell%20Growth%3A%20Targeting%20via%20Ketogenic%20Diet%20%26amp%3B%20Fasting%20ft.%20Dr.%20Keith%20Baar.%20HVMN%20Podcast.%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22podcast%22%2C%22title%22%3A%22mTor%20Signaling%20%26%20Cell%20Growth%3A%20Targeting%20via%20Ketogenic%20Diet%20%26%20Fasting%20ft.%20Dr.%20Keith%20Baar.%20HVMN%20Podcast.%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22podcaster%22%2C%22firstName%22%3A%22G.%22%2C%22lastName%22%3A%22Woo%22%7D%2C%7B%22creatorType%22%3A%22guest%22%2C%22firstName%22%3A%22K.%22%2C%22lastName%22%3A%22Baar%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22episodeNumber%22%3A%22%22%2C%22audioFileType%22%3A%22mp3%22%2C%22runningTime%22%3A%22%22%2C%22url%22%3A%22%22%2C%22language%22%3A%22%22%2C%22collections%22%3A%5B%22KY3SBXGN%22%5D%2C%22dateModified%22%3A%222019-07-18T02%3A46%3A32Z%22%7D%7D%2C%7B%22key%22%3A%2283UM7QL8%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22McCay%20et%20al.%22%2C%22parsedDate%22%3A%221935%22%2C%22numChildren%22%3A1%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EMcCay%20CM%2C%20Crowell%20MF%2C%20Maynard%20LA%20%281935%29%20The%20effect%20of%20retarded%20growth%20upon%20the%20length%20of%20life%20span%20and%20upon%20the%20ultimate%20body%20size.%20J%20Nutr%2010%3A63%26%23x2013%3B79.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1093%5C%2Fjn%5C%2F10.1.63%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1093%5C%2Fjn%5C%2F10.1.63%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22The%20effect%20of%20retarded%20growth%20upon%20the%20length%20of%20life%20span%20and%20upon%20the%20ultimate%20body%20size.%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22C.%20M.%22%2C%22lastName%22%3A%22McCay%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%20F.%22%2C%22lastName%22%3A%22Crowell%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22L.%20A.%22%2C%22lastName%22%3A%22Maynard%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%221935%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%2210.1093%5C%2Fjn%5C%2F10.1.63%22%2C%22ISSN%22%3A%22%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22A9H6NEVG%22%5D%2C%22dateModified%22%3A%222019-07-18T02%3A39%3A10Z%22%7D%7D%2C%7B%22key%22%3A%22JE5J8BEP%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Klass%22%2C%22parsedDate%22%3A%221977-12%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EKlass%20MR%20%281977%29%20Aging%20in%20the%20nematode%20Caenorhabditis%20elegans%3A%20major%20biological%20and%20environmental%20factors%20influencing%20life%20span.%20Mech%20Ageing%20Dev%206%3A413%26%23x2013%3B429.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2F0047-6374%2877%2990043-4%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2F0047-6374%2877%2990043-4%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Aging%20in%20the%20nematode%20Caenorhabditis%20elegans%3A%20major%20biological%20and%20environmental%20factors%20influencing%20life%20span%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%20R.%22%2C%22lastName%22%3A%22Klass%22%7D%5D%2C%22abstractNote%22%3A%22The%20free-living%20nematode%20Caenorhabditis%20elegans%20is%20an%20excellent%20experimental%20system%20for%20the%20study%20of%20aging.%20The%20present%20study%20identifies%20some%20of%20the%20major%20biological%20and%20environmental%20factors%20influencing%20life%20span%20as%20a%20prelude%20to%20more%20detailed%20genetic%20and%20biochemical%20analyses.%20Life%20span%20can%20be%20altered%20during%20any%20part%20of%20the%20life%20cycle%20by%20a%20change%20in%20either%20temperature%20or%20food%20concentration.%20Parental%20age%20and%20parental%20life%20span%20both%20have%20relatively%20small%20effects%20on%20progeny%20life%20span.%20The%20nematode%20accumulates%20fluorescent%20pigment%20resembling%20lipofuscin%2C%20and%20becomes%20less%20sensitive%20to%20ultra-violet%20radiation%20as%20it%20ages.%22%2C%22date%22%3A%221977%20Nov-Dec%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1016%5C%2F0047-6374%2877%2990043-4%22%2C%22ISSN%22%3A%220047-6374%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22A9H6NEVG%22%2C%22G7863IM7%22%5D%2C%22dateModified%22%3A%222019-07-18T02%3A38%3A17Z%22%7D%7D%2C%7B%22key%22%3A%22P2Z6B7GC%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Isley%20et%20al.%22%2C%22parsedDate%22%3A%221983-02%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EIsley%20WL%2C%20Underwood%20LE%2C%20Clemmons%20DR%20%281983%29%20Dietary%20components%20that%20regulate%20serum%20somatomedin-C%20concentrations%20in%20humans.%20J%20Clin%20Invest%2071%3A175%26%23x2013%3B182.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1172%5C%2Fjci110757%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1172%5C%2Fjci110757%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Dietary%20components%20that%20regulate%20serum%20somatomedin-C%20concentrations%20in%20humans%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22W.%20L.%22%2C%22lastName%22%3A%22Isley%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22L.%20E.%22%2C%22lastName%22%3A%22Underwood%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22D.%20R.%22%2C%22lastName%22%3A%22Clemmons%22%7D%5D%2C%22abstractNote%22%3A%22Dietary%20components%20responsible%20for%20the%20regulation%20of%20somatomedin-C%20in%20humans%20were%20assessed%20in%20five%20adult%20volunteers%20of%20normal%20weight%20who%20were%20fasted%20for%205%20d%20on%20three%20occasions%2C%20then%20refed%20three%20diets%20of%20differing%20composition.%20The%20serum%20somatomedin-C%20decreased%20from%20a%20mean%20prefasting%20value%20of%201.85%20%2B%5C%2F-%200.39%20U%5C%2Fml%20%28%2B%5C%2F-%201%20SD%29%20to%200.67%20%2B%5C%2F-%200.16%20U%5C%2Fml%20at%20the%20end%20of%20fasting%20%28P%20less%20than%200.005%29.%20After%20refeeding%20for%205%20d%20with%20a%20normal%20diet%2C%20the%20mean%20serum%20somatomedin-C%20increased%20to%201.26%20%2B%5C%2F-%200.20%20U%5C%2Fml.%20A%20protein-deficient%20%2832%25%20of%20control%29%2C%20isocaloric%20diet%20resulted%20in%20a%20significantly%20smaller%20increase%2C%20to%20a%20mean%20value%20of%200.90%20%2B%5C%2F-%200.24%20U%5C%2Fml%20%28P%20less%20than%200.05%29.%20A%20diet%20deficient%20in%20both%20protein%20and%20energy%20led%20to%20a%20further%20fall%200.31%20%2B%5C%2F-%200.06%20U%5C%2Fml.%20The%20changes%20in%20somatomedin-C%20during%20fasting%20and%20refeeding%20correlated%20significantly%20with%20mean%20daily%20nitrogen%20balance%20%28r%20%3D%200.90%29.%20We%20conclude%20that%20both%20protein%20and%20energy%20intake%20are%20regulators%20of%20serum%20somatomedin-C%20concentrations%20in%20adult%20humans%2C%20and%20energy%20intake%20may%20be%20of%20greater%20importance.%20The%20correlation%20between%20changes%20in%20somatomedin-C%20and%20nitrogen%20balance%20suggests%20that%20the%20former%20are%20directly%20related%20to%20changes%20in%20protein%20synthesis%20and%20may%20be%20helpful%20in%20assessing%20the%20response%20to%20nutritional%20therapy.%22%2C%22date%22%3A%22Feb%201983%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1172%5C%2Fjci110757%22%2C%22ISSN%22%3A%220021-9738%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22KMY5B6AS%22%2C%22XY9TFRAC%22%5D%2C%22dateModified%22%3A%222019-07-18T02%3A32%3A26Z%22%7D%7D%2C%7B%22key%22%3A%22AAWHJF5X%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Sahani%20et%20al.%22%2C%22parsedDate%22%3A%222014-03%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ESahani%20MH%2C%20Itakura%20E%2C%20Mizushima%20N%20%282014%29%20Expression%20of%20the%20autophagy%20substrate%20SQSTM1%5C%2Fp62%20is%20restored%20during%20prolonged%20starvation%20depending%20on%20transcriptional%20upregulation%20and%20autophagy-derived%20amino%20acids.%20Autophagy%2010%3A431%26%23x2013%3B441.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.4161%5C%2Fauto.27344%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.4161%5C%2Fauto.27344%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Expression%20of%20the%20autophagy%20substrate%20SQSTM1%5C%2Fp62%20is%20restored%20during%20prolonged%20starvation%20depending%20on%20transcriptional%20upregulation%20and%20autophagy-derived%20amino%20acids%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mayurbhai%20Himatbhai%22%2C%22lastName%22%3A%22Sahani%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eisuke%22%2C%22lastName%22%3A%22Itakura%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Noboru%22%2C%22lastName%22%3A%22Mizushima%22%7D%5D%2C%22abstractNote%22%3A%22SQSTM1%5C%2Fp62%20%28sequestosome%201%29%20is%20a%20multifunctional%20signaling%20molecule%2C%20involved%20in%20a%20variety%20of%20cellular%20pathways.%20SQSTM1%20is%20one%20of%20the%20best-known%20autophagic%20substrates%2C%20and%20is%20therefore%20widely%20used%20as%20an%20indicator%20of%20autophagic%20degradation.%20Here%20we%20report%20that%20the%20expression%20level%20of%20SQSTM1%20can%20be%20restored%20during%20prolonged%20starvation.%20Upon%20starvation%2C%20SQSTM1%20is%20initially%20degraded%20by%20autophagy.%20However%2C%20SQSTM1%20is%20restored%20back%20to%20basal%20levels%20during%20prolonged%20starvation%20in%20mouse%20embryonic%20fibroblasts%20and%20HepG2%20cells%2C%20but%20not%20in%20HeLa%20and%20HEK293%20cells.%20Restoration%20of%20SQSTM1%20depends%20on%20its%20transcriptional%20upregulation%2C%20which%20is%20triggered%20by%20amino%20acid%20starvation.%20Furthermore%2C%20amino%20acids%20derived%20from%20the%20autophagy-lysosome%20pathway%20are%20used%20for%20de%20novo%20synthesis%20of%20SQSTM1%20under%20starvation%20conditions.%20The%20restoration%20of%20SQSTM1%20is%20independent%20of%20reactivation%20of%20MTORC1%20%28mechanistic%20target%20of%20rapamycin%20complex%201%29.%20These%20results%20suggest%20that%20the%20expression%20level%20of%20SQSTM1%20in%20starved%20cells%20is%20determined%20by%20at%20least%203%20factors%3A%20autophagic%20degradation%2C%20transcriptional%20upregulation%2C%20and%20availability%20of%20lysosomal-derived%20amino%20acids.%20The%20results%20of%20this%20study%20also%20indicate%20that%20the%20expression%20level%20of%20SQSTM1%20does%20not%20always%20inversely%20correlate%20with%20autophagic%20activity.%22%2C%22date%22%3A%22Mar%202014%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.4161%5C%2Fauto.27344%22%2C%22ISSN%22%3A%221554-8635%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22FM3XX4QZ%22%5D%2C%22dateModified%22%3A%222019-07-17T00%3A17%3A36Z%22%7D%7D%2C%7B%22key%22%3A%22I2Y8YCZK%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Bj%5Cu00f8rk%5Cu00f8y%20et%20al.%22%2C%22parsedDate%22%3A%222005-11-21%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EBj%26%23xF8%3Brk%26%23xF8%3By%20G%2C%20Lamark%20T%2C%20Brech%20A%2C%20et%20al%20%282005%29%20p62%5C%2FSQSTM1%20forms%20protein%20aggregates%20degraded%20by%20autophagy%20and%20has%20a%20protective%20effect%20on%20huntingtin-induced%20cell%20death.%20J%20Cell%20Biol%20171%3A603%26%23x2013%3B614.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1083%5C%2Fjcb.200507002%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1083%5C%2Fjcb.200507002%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22p62%5C%2FSQSTM1%20forms%20protein%20aggregates%20degraded%20by%20autophagy%20and%20has%20a%20protective%20effect%20on%20huntingtin-induced%20cell%20death%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Geir%22%2C%22lastName%22%3A%22Bj%5Cu00f8rk%5Cu00f8y%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Trond%22%2C%22lastName%22%3A%22Lamark%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Andreas%22%2C%22lastName%22%3A%22Brech%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Heidi%22%2C%22lastName%22%3A%22Outzen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Maria%22%2C%22lastName%22%3A%22Perander%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Aud%22%2C%22lastName%22%3A%22%5Cu00d8vervatn%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Harald%22%2C%22lastName%22%3A%22Stenmark%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Terje%22%2C%22lastName%22%3A%22Johansen%22%7D%5D%2C%22abstractNote%22%3A%22Autophagic%20degradation%20of%20ubiquitinated%20protein%20aggregates%20is%20important%20for%20cell%20survival%2C%20but%20it%20is%20not%20known%20how%20the%20autophagic%20machinery%20recognizes%20such%20aggregates.%20In%20this%20study%2C%20we%20report%20that%20polymerization%20of%20the%20polyubiquitin-binding%20protein%20p62%5C%2FSQSTM1%20yields%20protein%20bodies%20that%20either%20reside%20free%20in%20the%20cytosol%20and%20nucleus%20or%20occur%20within%20autophagosomes%20and%20lysosomal%20structures.%20Inhibition%20of%20autophagy%20led%20to%20an%20increase%20in%20the%20size%20and%20number%20of%20p62%20bodies%20and%20p62%20protein%20levels.%20The%20autophagic%20marker%20light%20chain%203%20%28LC3%29%20colocalized%20with%20p62%20bodies%20and%20coimmunoprecipitated%20with%20p62%2C%20suggesting%20that%20these%20two%20proteins%20participate%20in%20the%20same%20complexes.%20The%20depletion%20of%20p62%20inhibited%20recruitment%20of%20LC3%20to%20autophagosomes%20under%20starvation%20conditions.%20Strikingly%2C%20p62%20and%20LC3%20formed%20a%20shell%20surrounding%20aggregates%20of%20mutant%20huntingtin.%20Reduction%20of%20p62%20protein%20levels%20or%20interference%20with%20p62%20function%20significantly%20increased%20cell%20death%20that%20was%20induced%20by%20the%20expression%20of%20mutant%20huntingtin.%20We%20suggest%20that%20p62%20may%2C%20via%20LC3%2C%20be%20involved%20in%20linking%20polyubiquitinated%20protein%20aggregates%20to%20the%20autophagy%20machinery.%22%2C%22date%22%3A%222005-11-21%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%2210.1083%5C%2Fjcb.200507002%22%2C%22ISSN%22%3A%220021-9525%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.ncbi.nlm.nih.gov%5C%2Fpmc%5C%2Farticles%5C%2FPMC2171557%5C%2F%22%2C%22collections%22%3A%5B%22FM3XX4QZ%22%5D%2C%22dateModified%22%3A%222019-07-17T00%3A11%3A56Z%22%7D%7D%2C%7B%22key%22%3A%22HRIQWFPR%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Epstein%20et%20al.%22%2C%22parsedDate%22%3A%222017%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EEpstein%20T%2C%20Gatenby%20RA%2C%20Brown%20JS%20%282017%29%20The%20Warburg%20effect%20as%20an%20adaptation%20of%20cancer%20cells%20to%20rapid%20fluctuations%20in%20energy%20demand.%20PLoS%20ONE%2012%3Ae0185085.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1371%5C%2Fjournal.pone.0185085%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1371%5C%2Fjournal.pone.0185085%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22The%20Warburg%20effect%20as%20an%20adaptation%20of%20cancer%20cells%20to%20rapid%20fluctuations%20in%20energy%20demand%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tamir%22%2C%22lastName%22%3A%22Epstein%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Robert%20A.%22%2C%22lastName%22%3A%22Gatenby%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Joel%20S.%22%2C%22lastName%22%3A%22Brown%22%7D%5D%2C%22abstractNote%22%3A%22To%20maintain%20optimal%20fitness%2C%20a%20cell%20must%20balance%20the%20risk%20of%20inadequate%20energy%20reserve%20for%20response%20to%20a%20potentially%20fatal%20perturbation%20against%20the%20long-term%20cost%20of%20maintaining%20high%20concentrations%20of%20ATP%20to%20meet%20occasional%20spikes%20in%20demand.%20Here%20we%20apply%20a%20game%20theoretic%20approach%20to%20address%20the%20dynamics%20of%20energy%20production%20and%20expenditure%20in%20eukaryotic%20cells.%20Conventionally%2C%20glucose%20metabolism%20is%20viewed%20as%20a%20function%20of%20oxygen%20concentrations%20in%20which%20the%20more%20efficient%20oxidation%20of%20glucose%20to%20CO2%20and%20H2O%20produces%20all%20or%20nearly%20all%20ATP%20except%20under%20hypoxic%20conditions%20when%20less%20efficient%20%282%20ATP%5C%2F%20glucose%20vs.%20about%2036ATP%5C%2Fglucose%29%20anaerobic%20metabolism%20of%20glucose%20to%20lactic%20acid%20provides%20an%20emergency%20backup.%20We%20propose%20an%20alternative%20in%20which%20energy%20production%20is%20governed%20by%20the%20complex%20temporal%20and%20spatial%20dynamics%20of%20intracellular%20ATP%20demand.%20In%20the%20short%20term%2C%20a%20cell%20must%20provide%20energy%20for%20constant%20baseline%20needs%20but%20also%20maintain%20capacity%20to%20rapidly%20respond%20to%20fluxes%20in%20demand%20particularly%20due%20to%20external%20perturbations%20on%20the%20cell%20membrane.%20Similarly%2C%20longer-term%20dynamics%20require%20a%20trade-off%20between%20the%20cost%20of%20maintaining%20high%20metabolic%20capacity%20to%20meet%20uncommon%20spikes%20in%20demand%20versus%20the%20risk%20of%20unsuccessfully%20responding%20to%20threats%20or%20opportunities.%20Here%20we%20develop%20a%20model%20and%20computationally%20explore%20the%20cell%27s%20optimal%20mix%20of%20glycolytic%20and%20oxidative%20capacity.%20We%20find%20the%20Warburg%20effect%2C%20high%20glycolytic%20metabolism%20even%20under%20normoxic%20conditions%2C%20is%20represents%20a%20metabolic%20strategy%20that%20allow%20cancer%20cells%20to%20optimally%20meet%20energy%20demands%20posed%20by%20stochastic%20or%20fluctuating%20tumor%20environments.%22%2C%22date%22%3A%222017%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1371%5C%2Fjournal.pone.0185085%22%2C%22ISSN%22%3A%221932-6203%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22VUGC9HHJ%22%5D%2C%22dateModified%22%3A%222019-07-15T00%3A29%3A43Z%22%7D%7D%2C%7B%22key%22%3A%22UMACU7KS%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Seyfried%20et%20al.%22%2C%22parsedDate%22%3A%222014-03%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ESeyfried%20TN%2C%20Flores%20RE%2C%20Poff%20AM%2C%20D%26%23x2019%3BAgostino%20DP%20%282014%29%20Cancer%20as%20a%20metabolic%20disease%3A%20implications%20for%20novel%20therapeutics.%20Carcinogenesis%2035%3A515%26%23x2013%3B527.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1093%5C%2Fcarcin%5C%2Fbgt480%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1093%5C%2Fcarcin%5C%2Fbgt480%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Cancer%20as%20a%20metabolic%20disease%3A%20implications%20for%20novel%20therapeutics%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Thomas%20N.%22%2C%22lastName%22%3A%22Seyfried%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Roberto%20E.%22%2C%22lastName%22%3A%22Flores%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Angela%20M.%22%2C%22lastName%22%3A%22Poff%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Dominic%20P.%22%2C%22lastName%22%3A%22D%5Cu2019Agostino%22%7D%5D%2C%22abstractNote%22%3A%22Emerging%20evidence%20indicates%20that%20cancer%20is%20primarily%20a%20metabolic%20disease%20involving%20disturbances%20in%20energy%20production%20through%20respiration%20and%20fermentation.%20The%20genomic%20instability%20observed%20in%20tumor%20cells%20and%20all%20other%20recognized%20hallmarks%20of%20cancer%20are%20considered%20downstream%20epiphenomena%20of%20the%20initial%20disturbance%20of%20cellular%20energy%20metabolism.%20The%20disturbances%20in%20tumor%20cell%20energy%20metabolism%20can%20be%20linked%20to%20abnormalities%20in%20the%20structure%20and%20function%20of%20the%20mitochondria.%20When%20viewed%20as%20a%20mitochondrial%20metabolic%20disease%2C%20the%20evolutionary%20theory%20of%20Lamarck%20can%20better%20explain%20cancer%20progression%20than%20can%20the%20evolutionary%20theory%20of%20Darwin.%20Cancer%20growth%20and%20progression%20can%20be%20managed%20following%20a%20whole%20body%20transition%20from%20fermentable%20metabolites%2C%20primarily%20glucose%20and%20glutamine%2C%20to%20respiratory%20metabolites%2C%20primarily%20ketone%20bodies.%20As%20each%20individual%20is%20a%20unique%20metabolic%20entity%2C%20personalization%20of%20metabolic%20therapy%20as%20a%20broad-based%20cancer%20treatment%20strategy%20will%20require%20fine-tuning%20to%20match%20the%20therapy%20to%20an%20individual%5Cu2019s%20unique%20physiology.%22%2C%22date%22%3A%222014-3%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%2210.1093%5C%2Fcarcin%5C%2Fbgt480%22%2C%22ISSN%22%3A%220143-3334%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.ncbi.nlm.nih.gov%5C%2Fpmc%5C%2Farticles%5C%2FPMC3941741%5C%2F%22%2C%22collections%22%3A%5B%22VUGC9HHJ%22%2C%22QZ2YBDFE%22%5D%2C%22dateModified%22%3A%222019-07-14T23%3A44%3A27Z%22%7D%7D%2C%7B%22key%22%3A%22PWKABTDZ%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Seyfried%22%2C%22parsedDate%22%3A%222015-07-07%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ESeyfried%20TN%20%282015%29%20Cancer%20as%20a%20mitochondrial%20metabolic%20disease.%20Front%20Cell%20Dev%20Biol%203%3A.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3389%5C%2Ffcell.2015.00043%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3389%5C%2Ffcell.2015.00043%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Cancer%20as%20a%20mitochondrial%20metabolic%20disease%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Thomas%20N.%22%2C%22lastName%22%3A%22Seyfried%22%7D%5D%2C%22abstractNote%22%3A%22Cancer%20is%20widely%20considered%20a%20genetic%20disease%20involving%20nuclear%20mutations%20in%20oncogenes%20and%20tumor%20suppressor%20genes.%20This%20view%20persists%20despite%20the%20numerous%20inconsistencies%20associated%20with%20the%20somatic%20mutation%20theory.%20In%20contrast%20to%20the%20somatic%20mutation%20theory%2C%20emerging%20evidence%20suggests%20that%20cancer%20is%20a%20mitochondrial%20metabolic%20disease%2C%20according%20to%20the%20original%20theory%20of%20Otto%20Warburg.%20The%20findings%20are%20reviewed%20from%20nuclear%20cytoplasm%20transfer%20experiments%20that%20relate%20to%20the%20origin%20of%20cancer.%20The%20evidence%20from%20these%20experiments%20is%20difficult%20to%20reconcile%20with%20the%20somatic%20mutation%20theory%2C%20but%20is%20consistent%20with%20the%20notion%20that%20cancer%20is%20primarily%20a%20mitochondrial%20metabolic%20disease.%22%2C%22date%22%3A%222015-7-07%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%2210.3389%5C%2Ffcell.2015.00043%22%2C%22ISSN%22%3A%222296-634X%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.ncbi.nlm.nih.gov%5C%2Fpmc%5C%2Farticles%5C%2FPMC4493566%5C%2F%22%2C%22collections%22%3A%5B%22VUGC9HHJ%22%5D%2C%22dateModified%22%3A%222019-07-14T23%3A41%3A58Z%22%7D%7D%2C%7B%22key%22%3A%22MDJEWPB4%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Brahmkhatri%20et%20al.%22%2C%22parsedDate%22%3A%222015%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EBrahmkhatri%20VP%2C%20Prasanna%20C%2C%20Atreya%20HS%20%282015%29%20Insulin-Like%20Growth%20Factor%20System%20in%20Cancer%3A%20Novel%20Targeted%20Therapies.%20Biomed%20Res%20Int%202015%3A.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1155%5C%2F2015%5C%2F538019%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1155%5C%2F2015%5C%2F538019%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Insulin-Like%20Growth%20Factor%20System%20in%20Cancer%3A%20Novel%20Targeted%20Therapies%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Varsha%20P.%22%2C%22lastName%22%3A%22Brahmkhatri%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Chinmayi%22%2C%22lastName%22%3A%22Prasanna%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hanudatta%20S.%22%2C%22lastName%22%3A%22Atreya%22%7D%5D%2C%22abstractNote%22%3A%22Insulin-like%20growth%20factors%20%28IGFs%29%20are%20essential%20for%20growth%20and%20survival%20that%20suppress%20apoptosis%20and%20promote%20cell%20cycle%20progression%2C%20angiogenesis%2C%20and%20metastatic%20activities%20in%20various%20cancers.%20The%20IGFs%20actions%20are%20mediated%20through%20the%20IGF-1%20receptor%20that%20is%20involved%20in%20cell%20transformation%20induced%20by%20tumour.%20These%20effects%20depend%20on%20the%20bioavailability%20of%20IGFs%2C%20which%20is%20regulated%20by%20IGF%20binding%20proteins%20%28IGFBPs%29.%20We%20describe%20here%20the%20role%20of%20the%20IGF%20system%20in%20cancer%2C%20proposing%20new%20strategies%20targeting%20this%20system.%20We%20have%20attempted%20to%20expand%20the%20general%20viewpoint%20on%20IGF-1R%2C%20its%20inhibitors%2C%20potential%20limitations%20of%20IGF-1R%2C%20antibodies%20and%20tyrosine%20kinase%20inhibitors%2C%20and%20IGFBP%20actions.%20This%20review%20discusses%20the%20emerging%20view%20that%20blocking%20IGF%20via%20IGFBP%20is%20a%20better%20option%20than%20blocking%20IGF%20receptors.%20This%20can%20lead%20to%20the%20development%20of%20novel%20cancer%20therapies.%22%2C%22date%22%3A%222015%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%2210.1155%5C%2F2015%5C%2F538019%22%2C%22ISSN%22%3A%222314-6133%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.ncbi.nlm.nih.gov%5C%2Fpmc%5C%2Farticles%5C%2FPMC4383470%5C%2F%22%2C%22collections%22%3A%5B%22NBR5UDJK%22%5D%2C%22dateModified%22%3A%222019-07-14T23%3A27%3A53Z%22%7D%7D%2C%7B%22key%22%3A%22L5K2CMYB%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22He%20et%20al.%22%2C%22parsedDate%22%3A%222012-01-18%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EHe%20C%2C%20Bassik%20MC%2C%20Moresi%20V%2C%20et%20al%20%282012%29%20Exercise%26%23x2013%3Binduced%20BCL2%26%23x2013%3Bregulated%20autophagy%20is%20required%20for%20muscle%20glucose%20homeostasis.%20Nature%20481%3A511%26%23x2013%3B515.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1038%5C%2Fnature10758%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1038%5C%2Fnature10758%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Exercise%5Cu2013induced%20BCL2%5Cu2013regulated%20autophagy%20is%20required%20for%20muscle%20glucose%20homeostasis%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Congcong%22%2C%22lastName%22%3A%22He%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Michael%20C.%22%2C%22lastName%22%3A%22Bassik%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Viviana%22%2C%22lastName%22%3A%22Moresi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kai%22%2C%22lastName%22%3A%22Sun%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yongjie%22%2C%22lastName%22%3A%22Wei%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Zhongju%22%2C%22lastName%22%3A%22Zou%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Zhenyi%22%2C%22lastName%22%3A%22An%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Joy%22%2C%22lastName%22%3A%22Loh%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jill%22%2C%22lastName%22%3A%22Fisher%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Qihua%22%2C%22lastName%22%3A%22Sun%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Stanley%22%2C%22lastName%22%3A%22Korsmeyer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Milton%22%2C%22lastName%22%3A%22Packer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Herman%20I.%22%2C%22lastName%22%3A%22May%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Joseph%20A.%22%2C%22lastName%22%3A%22Hill%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Herbert%20W.%22%2C%22lastName%22%3A%22Virgin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christopher%22%2C%22lastName%22%3A%22Gilpin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guanghua%22%2C%22lastName%22%3A%22Xiao%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Rhonda%22%2C%22lastName%22%3A%22Bassel-Duby%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Philipp%20E.%22%2C%22lastName%22%3A%22Scherer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Beth%22%2C%22lastName%22%3A%22Levine%22%7D%5D%2C%22abstractNote%22%3A%22Exercise%20has%20beneficial%20effects%20on%20human%20health%2C%20including%20protection%20against%20metabolic%20disorders%20such%20as%20diabetes.%20However%2C%20the%20cellular%20mechanisms%20underlying%20these%20effects%20are%20incompletely%20understood.%20The%20lysosomal%20degradation%20pathway%2C%20autophagy%2C%20is%20an%20intracellular%20recycling%20system%20that%20functions%20during%20basal%20conditions%20in%20organelle%20and%20protein%20quality%20control.%20During%20stress%2C%20increased%20levels%20of%20autophagy%20permit%20cells%20to%20adapt%20to%20changing%20nutritional%20and%20energy%20demands%20through%20protein%20catabolism.%20Moreover%2C%20in%20animal%20models%2C%20autophagy%20protects%20against%20diseases%20such%20as%20cancer%2C%20neuro-degenerative%20disorders%2C%20infections%2C%20inflammatory%20diseases%2C%20ageing%20and%20insulin%20resistance-.%20Here%20we%20show%20that%20acute%20exercise%20induces%20autophagy%20in%20skeletal%20and%20cardiac%20muscle%20of%20fed%20mice.%20To%20investigate%20the%20role%20of%20exercise-mediated%20autophagy%20in%20vivo%2C%20we%20generated%20mutant%20mice%20that%20show%20normal%20levels%20of%20basal%20autophagy%20but%20are%20deficient%20in%20stimulus%20%28exercise-%20or%20starvation%29-induced%20autophagy.%20These%20mice%20%28termed%20BCL2%20AAA%20mice%29%20contain%20knock-in%20mutations%20in%20BCL2%20phosphorylation%20sites%20%28Thr69Ala%2C%20Ser70Ala%20and%20Ser84Ala%29%20that%20prevent%20stimulus-induced%20disruption%20of%20the%20BCL2-beclin-1%20complex%20and%20autophagy%20activation.%20BCL2%20AAA%20mice%20show%20decreased%20endurance%20and%20altered%20glucose%20metabolism%20during%20acute%20exercise%2C%20as%20well%20as%20impaired%20chronic%20exercise-mediated%20protection%20against%20high-fat-diet-induced%20glucose%20intolerance.%20Thus%2C%20exercise%20induces%20autophagy%2C%20BCL2%20is%20a%20crucial%20regulator%20of%20exercise-%20%28and%20starvation%29-%20induced%20autophagy%20in%20vivo%2C%20and%20autophagy%20induction%20may%20contribute%20to%20the%20beneficial%20metabolic%20effects%20of%20exercise.%22%2C%22date%22%3A%222012-1-18%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%2210.1038%5C%2Fnature10758%22%2C%22ISSN%22%3A%220028-0836%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.ncbi.nlm.nih.gov%5C%2Fpmc%5C%2Farticles%5C%2FPMC3518436%5C%2F%22%2C%22collections%22%3A%5B%22FM3XX4QZ%22%5D%2C%22dateModified%22%3A%222019-07-14T23%3A08%3A33Z%22%7D%7D%2C%7B%22key%22%3A%22CUMUP9TI%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Marinac%20et%20al.%22%2C%22parsedDate%22%3A%222016-08-01%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EMarinac%20CR%2C%20Nelson%20SH%2C%20Breen%20CI%2C%20et%20al%20%282016%29%20Prolonged%20Nightly%20Fasting%20and%20Breast%20Cancer%20Prognosis.%20JAMA%20Oncol%202%3A1049%26%23x2013%3B1055.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1001%5C%2Fjamaoncol.2016.0164%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1001%5C%2Fjamaoncol.2016.0164%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Prolonged%20Nightly%20Fasting%20and%20Breast%20Cancer%20Prognosis%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Catherine%20R.%22%2C%22lastName%22%3A%22Marinac%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sandahl%20H.%22%2C%22lastName%22%3A%22Nelson%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Caitlin%20I.%22%2C%22lastName%22%3A%22Breen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sheri%20J.%22%2C%22lastName%22%3A%22Hartman%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Loki%22%2C%22lastName%22%3A%22Natarajan%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22John%20P.%22%2C%22lastName%22%3A%22Pierce%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Shirley%20W.%22%2C%22lastName%22%3A%22Flatt%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Dorothy%20D.%22%2C%22lastName%22%3A%22Sears%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ruth%20E.%22%2C%22lastName%22%3A%22Patterson%22%7D%5D%2C%22abstractNote%22%3A%22IMPORTANCE%5CnRodent%20studies%20demonstrate%20that%20prolonged%20fasting%20during%20the%20sleep%20phase%20positively%20influences%20carcinogenesis%20and%20metabolic%20processes%20that%20are%20putatively%20associated%20with%20risk%20and%20prognosis%20of%20breast%20cancer.%20To%20our%20knowledge%2C%20no%20studies%20in%20humans%20have%20examined%20nightly%20fasting%20duration%20and%20cancer%20outcomes.%5Cn%5CnOBJECTIVE%5CnTo%20investigate%20whether%20duration%20of%20nightly%20fasting%20predicted%20recurrence%20and%20mortality%20among%20women%20with%20early-stage%20breast%20cancer%20and%2C%20if%20so%2C%20whether%20it%20was%20associated%20with%20risk%20factors%20for%20poor%20outcomes%2C%20including%20glucoregulation%20%28hemoglobin%20A1c%29%2C%20chronic%20inflammation%20%28C-reactive%20protein%29%2C%20obesity%2C%20and%20sleep.%5Cn%5CnDESIGN%2C%20SETTING%2C%20AND%20PARTICIPANTS%5CnData%20were%20collected%20from%202413%20women%20with%20breast%20cancer%20but%20without%20diabetes%20mellitus%20who%20were%20aged%2027%20to%2070%20years%20at%20diagnosis%20and%20participated%20in%20the%20prospective%20Women%5Cu2019s%20Healthy%20Eating%20and%20Living%20study%20between%20March%201%2C%201995%2C%20and%20May%203%2C%202007.%20Data%20analysis%20was%20conducted%20from%20May%2018%20to%20October%205%2C%202015.%5Cn%5CnEXPOSURES%5CnNightly%20fasting%20duration%20was%20estimated%20from%2024-hour%20dietary%20recalls%20collected%20at%20baseline%2C%20year%201%2C%20and%20year%204.%5Cn%5CnMAIN%20OUTCOMES%20AND%20MEASURES%5CnClinical%20outcomes%20were%20invasive%20breast%20cancer%20recurrence%20and%20new%20primary%20breast%20tumors%20during%20a%20mean%20of%207.3%20years%20of%20study%20follow-up%20as%20well%20as%20death%20from%20breast%20cancer%20or%20any%20cause%20during%20a%20mean%20of%2011.4%20years%20of%20surveillance.%20Baseline%20sleep%20duration%20was%20self-reported%2C%20and%20archived%20blood%20samples%20were%20used%20to%20assess%20concentrations%20of%20hemoglobin%20A1c%20and%20C-reactive%20protein.%5Cn%5CnRESULTS%5CnThe%20cohort%20of%202413%20women%20%28mean%20%5BSD%5D%20age%2C%2052.4%20%5B8.9%5D%20years%29%20reported%20a%20mean%20%28SD%29%20fasting%20duration%20of%2012.5%20%281.7%29%20hours%20per%20night.%20In%20repeated-measures%20Cox%20proportional%20hazards%20regression%20models%2C%20fasting%20less%20than%2013%20hours%20per%20night%20%28lower%202%20tertiles%20of%20nightly%20fasting%20distribution%29%20was%20associated%20with%20an%20increase%20in%20the%20risk%20of%20breast%20cancer%20recurrence%20compared%20with%20fasting%2013%20or%20more%20hours%20per%20night%20%28hazard%20ratio%2C%201.36%3B%2095%25%20CI%2C%201.05-1.76%29.%20Nightly%20fasting%20less%20than%2013%20hours%20was%20not%20associated%20with%20a%20statistically%20significant%20higher%20risk%20of%20breast%20cancer%20mortality%20%28hazard%20ratio%2C%201.21%3B%2095%25%20CI%2C%200.91-1.60%29%20or%20a%20statistically%20significant%20higher%20risk%20of%20all-cause%20mortality%20%28hazard%20ratio%2C%201.22%3B%2095%25%20CI%2C%200.95-1.56%29.%20In%20multivariable%20linear%20regression%20models%2C%20each%202-hour%20increase%20in%20the%20nightly%20fasting%20duration%20was%20associated%20with%20significantly%20lower%20hemoglobin%20A1c%20levels%20%28%5Cu03b2%20%3D%20%5Cu22120.37%3B%2095%25%20CI%2C%20%5Cu22120.72%20to%20%5Cu22120.01%29%20and%20a%20longer%20duration%20of%20nighttime%20sleep%20%28%5Cu03b2%20%3D%200.20%3B%2095%25%20CI%2C%200.14-0.26%29.%5Cn%5CnCONCLUSIONS%20AND%20RELEVANCE%5CnProlonging%20the%20length%20of%20the%20nightly%20fasting%20interval%20may%20be%20a%20simple%2C%20nonpharmacologic%20strategy%20for%20reducing%20the%20risk%20of%20breast%20cancer%20recurrence.%20Improvements%20in%20glucoregulation%20and%20sleep%20may%20be%20mechanisms%20linking%20nightly%20fasting%20with%20breast%20cancer%20prognosis.%22%2C%22date%22%3A%222016-8-1%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%2210.1001%5C%2Fjamaoncol.2016.0164%22%2C%22ISSN%22%3A%222374-2437%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.ncbi.nlm.nih.gov%5C%2Fpmc%5C%2Farticles%5C%2FPMC4982776%5C%2F%22%2C%22collections%22%3A%5B%22UIM8YXRX%22%2C%22UP98JRCY%22%5D%2C%22dateModified%22%3A%222019-07-14T21%3A04%3A04Z%22%7D%7D%2C%7B%22key%22%3A%22E6IMUMCA%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Clemmons%20et%20al.%22%2C%22parsedDate%22%3A%221981-12%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EClemmons%20DR%2C%20Klibanski%20A%2C%20Underwood%20LE%2C%20et%20al%20%281981%29%20Reduction%20of%20plasma%20immunoreactive%20somatomedin%20C%20during%20fasting%20in%20humans.%20J%20Clin%20Endocrinol%20Metab%2053%3A1247%26%23x2013%3B1250.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1210%5C%2Fjcem-53-6-1247%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1210%5C%2Fjcem-53-6-1247%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Reduction%20of%20plasma%20immunoreactive%20somatomedin%20C%20during%20fasting%20in%20humans%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22D.%20R.%22%2C%22lastName%22%3A%22Clemmons%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%22%2C%22lastName%22%3A%22Klibanski%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22L.%20E.%22%2C%22lastName%22%3A%22Underwood%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22J.%20W.%22%2C%22lastName%22%3A%22McArthur%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22E.%20C.%22%2C%22lastName%22%3A%22Ridgway%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22I.%20Z.%22%2C%22lastName%22%3A%22Beitins%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22J.%20J.%22%2C%22lastName%22%3A%22Van%20Wyk%22%7D%5D%2C%22abstractNote%22%3A%22We%20have%20assessed%20the%20effect%20of%20fasting%20for%2010%20days%20on%20plasma%20concentrations%20of%20immunoreactive%20somatomedin%20C%20and%20urinary%20urea%20excretion%20in%20seven%20obese%20male%20volunteers.%20From%20a%20mean%20prefast%20value%20of%200.83%20U%5C%2Fml%2C%20plasma%20somatomedin%20C%20fell%20to%200.21%20U%5C%2Fml%20after%2010%20days%20of%20fasting.%20A%20prompt%20increase%20was%20observed%20with%20refeeding.%20The%20change%20in%20somatomedin%20C%20during%20fasting%20showed%20a%20highly%20significant%20correlation%20with%20the%20change%20in%20urinary%20urea%20nitrogen%20excretion%20%28r%20%3D%200.74%3B%20P%20less%20than%200.001%29.%20It%20also%20was%20shown%20that%20inhibitors%20which%20interfere%20with%20quantitation%20in%20somatomedin%20bioassays%20are%20not%20observed%20in%20the%20RIA%20for%20somatomedin%20C.%20The%20results%20of%20this%20study%20suggest%20that%20measurement%20of%20plasma%20somatomedin%20C%20provides%20a%20sensitive%20indicator%20of%20nitrogen%20loss%20and%20may%20be%20useful%20in%20monitoring%20the%20changes%20in%20protein%20metabolism%20that%20occur%20during%20alterations%20in%20nutritional%20status.%22%2C%22date%22%3A%22Dec%201981%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1210%5C%2Fjcem-53-6-1247%22%2C%22ISSN%22%3A%220021-972X%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22KMY5B6AS%22%2C%22XY9TFRAC%22%5D%2C%22dateModified%22%3A%222019-07-14T02%3A23%3A52Z%22%7D%7D%2C%7B%22key%22%3A%224XGWAUUR%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Yoshii%20and%20Mizushima%22%2C%22parsedDate%22%3A%222017-08-28%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EYoshii%20SR%2C%20Mizushima%20N%20%282017%29%20Monitoring%20and%20Measuring%20Autophagy.%20Int%20J%20Mol%20Sci%2018%3A.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3390%5C%2Fijms18091865%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3390%5C%2Fijms18091865%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Monitoring%20and%20Measuring%20Autophagy%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Saori%20R.%22%2C%22lastName%22%3A%22Yoshii%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Noboru%22%2C%22lastName%22%3A%22Mizushima%22%7D%5D%2C%22abstractNote%22%3A%22Autophagy%20is%20a%20cytoplasmic%20degradation%20system%2C%20which%20is%20important%20for%20starvation%20adaptation%20and%20cellular%20quality%20control.%20Recent%20advances%20in%20understanding%20autophagy%20highlight%20its%20importance%20under%20physiological%20and%20pathological%20conditions.%20However%2C%20methods%20for%20monitoring%20autophagic%20activity%20are%20complicated%20and%20the%20results%20are%20sometimes%20misinterpreted.%20Here%2C%20we%20review%20the%20methods%20used%20to%20identify%20autophagic%20structures%2C%20and%20to%20measure%20autophagic%20flux%20in%20cultured%20cells%20and%20animals.%20We%20will%20also%20describe%20the%20existing%20autophagy%20reporter%20mice%20that%20are%20useful%20for%20autophagy%20studies%20and%20drug%20testing.%20Lastly%2C%20we%20will%20consider%20the%20attempts%20to%20monitor%20autophagy%20in%20samples%20derived%20from%20humans.%22%2C%22date%22%3A%222017-8-28%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%2210.3390%5C%2Fijms18091865%22%2C%22ISSN%22%3A%221422-0067%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.ncbi.nlm.nih.gov%5C%2Fpmc%5C%2Farticles%5C%2FPMC5618514%5C%2F%22%2C%22collections%22%3A%5B%22SBXV6KNU%22%5D%2C%22dateModified%22%3A%222019-07-14T01%3A07%3A37Z%22%7D%7D%2C%7B%22key%22%3A%22ZGACAGRI%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Pietrocola%20et%20al.%22%2C%22parsedDate%22%3A%222014-06-15%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EPietrocola%20F%2C%20Malik%20SA%2C%20Mari%26%23xF1%3Bo%20G%2C%20et%20al%20%282014%29%20Coffee%20induces%20autophagy%20in%20vivo.%20Cell%20Cycle%2013%3A1987%26%23x2013%3B1994.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.4161%5C%2Fcc.28929%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.4161%5C%2Fcc.28929%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Coffee%20induces%20autophagy%20in%20vivo%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Federico%22%2C%22lastName%22%3A%22Pietrocola%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Shoaib%20Ahmad%22%2C%22lastName%22%3A%22Malik%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guillermo%22%2C%22lastName%22%3A%22Mari%5Cu00f1o%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Erika%22%2C%22lastName%22%3A%22Vacchelli%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Laura%22%2C%22lastName%22%3A%22Senovilla%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kariman%22%2C%22lastName%22%3A%22Chaba%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mireia%22%2C%22lastName%22%3A%22Niso-Santano%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Maria%20Chiara%22%2C%22lastName%22%3A%22Maiuri%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Frank%22%2C%22lastName%22%3A%22Madeo%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Kroemer%22%7D%5D%2C%22abstractNote%22%3A%22Epidemiological%20studies%20and%20clinical%20trials%20revealed%20that%20chronic%20consumption%20coffee%20is%20associated%20with%20the%20inhibition%20of%20several%20metabolic%20diseases%20as%20well%20as%20reduction%20in%20overall%20and%20cause-specific%20mortality.%20We%20show%20that%20both%20natural%20and%20decaffeinated%20brands%20of%20coffee%20similarly%20rapidly%20trigger%20autophagy%20in%20mice.%20One%20to%204%20h%20after%20coffee%20consumption%2C%20we%20observed%20an%20increase%20in%20autophagic%20flux%20in%20all%20investigated%20organs%20%28liver%2C%20muscle%2C%20heart%29%20in%20vivo%2C%20as%20indicated%20by%20the%20increased%20lipidation%20of%20LC3B%20and%20the%20reduction%20of%20the%20abundance%20of%20the%20autophagic%20substrate%20sequestosome%201%20%28p62%5C%2FSQSTM1%29.%20These%20changes%20were%20accompanied%20by%20the%20inhibition%20of%20the%20enzymatic%20activity%20of%20mammalian%20target%20of%20rapamycin%20complex%201%20%28mTORC1%29%2C%20leading%20to%20the%20reduced%20phosphorylation%20of%20p70S6K%2C%20as%20well%20as%20by%20the%20global%20deacetylation%20of%20cellular%20proteins%20detectable%20by%20immunoblot.%20Immunohistochemical%20analyses%20of%20transgenic%20mice%20expressing%20a%20GFP%5Cu2013LC3B%20fusion%20protein%20confirmed%20the%20coffee-induced%20relocation%20of%20LC3B%20to%20autophagosomes%2C%20as%20well%20as%20general%20protein%20deacetylation.%20Altogether%2C%20these%20results%20indicate%20that%20coffee%20triggers%202%20phenomena%20that%20are%20also%20induced%20by%20nutrient%20depletion%2C%20namely%20a%20reduction%20of%20protein%20acetylation%20coupled%20to%20an%20increase%20in%20autophagy.%20We%20speculate%20that%20polyphenols%20contained%20in%20coffee%20promote%20health%20by%20stimulating%20autophagy.%22%2C%22date%22%3A%222014-6-15%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%2210.4161%5C%2Fcc.28929%22%2C%22ISSN%22%3A%221538-4101%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.ncbi.nlm.nih.gov%5C%2Fpmc%5C%2Farticles%5C%2FPMC4111762%5C%2F%22%2C%22collections%22%3A%5B%22FM3XX4QZ%22%5D%2C%22dateModified%22%3A%222019-07-12T13%3A13%3A07Z%22%7D%7D%2C%7B%22key%22%3A%226NCTSX67%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22de%20Haan%20et%20al.%22%2C%22parsedDate%22%3A%221997-03-01%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3Ede%20Haan%20G%2C%20Nijhof%20W%2C%20Van%20Zant%20G%20%281997%29%20Mouse%20strain-dependent%20changes%20in%20frequency%20and%20proliferation%20of%20hematopoietic%20stem%20cells%20during%20aging%3A%20correlation%20between%20lifespan%20and%20cycling%20activity.%20Blood%2089%3A1543%26%23x2013%3B1550%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Mouse%20strain-dependent%20changes%20in%20frequency%20and%20proliferation%20of%20hematopoietic%20stem%20cells%20during%20aging%3A%20correlation%20between%20lifespan%20and%20cycling%20activity%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22G.%22%2C%22lastName%22%3A%22de%20Haan%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22W.%22%2C%22lastName%22%3A%22Nijhof%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22G.%22%2C%22lastName%22%3A%22Van%20Zant%22%7D%5D%2C%22abstractNote%22%3A%22We%20have%20quantified%20the%20frequency%20and%20proliferation%20of%20five%20subsets%20of%20primitive%20hematopoietic%20cells%2C%20using%20the%20cobblestone%20area%20forming%20cell%20%28CAFC%29%20assay%2C%20in%20marrow%20of%20five%20strains%20of%20mice%20with%20lifespans%20ranging%20from%20about%20500%20to%20800%20days.%20Stem%20cell%20characteristics%20were%20determined%20in%20young%20%286%20weeks%29%20and%20old%20%2812%20months%29%20mice.%20We%20report%20striking%20effects%20of%20both%20intrinsic%20strain%20lifespan%20and%20individual%20mouse%20age%20on%20stem%20cell%20populations.%20First%2C%20the%20relative%20and%20absolute%20numbers%20of%20the%20most%20primitive%20stem%20cell%20subsets%20was%20threefold%20to%20fourfold%20higher%20in%20old%20than%20in%20young%20mice.%20Second%2C%20a%20considerable%20strain-to-strain%20variation%20in%20the%20number%20of%20primitive%20cells%20was%20observed%3A%20when%20absolute%20frequencies%20were%20calculated%2C%20there%20was%20a%20trend%20for%20longer%20lifespan%20to%20be%20correlated%20with%20a%20larger%20stem%20cell%20pool.%20Third%2C%20stem%20cells%20from%20old%20mice%20had%20a%20far%20lower%20cycling%20activity%20than%20cells%20from%20young%20mice.%20However%2C%20this%20was%20highly%20strain%20dependent%3A%20short-lived%20C3H%5C%2FHe%20and%20CBA%5C%2FJ%20mice%20showed%20a%20stronger%20reduction%20in%20cycling%20activity%20during%20aging%20than%20long-lived%20C57BL%5C%2F6%20mice.%20Finally%2C%20a%20significant%20negative%20correlation%20was%20demonstrated%20in%20young%20mice%20between%20maximal%20lifespan%20and%20proliferative%20activity.%20These%20data%20show%20that%20aging%20has%20a%20major%20impact%20on%20the%20frequency%20and%20cell-cycle%20kinetics%20of%20primitive%20hematopoietic%20cell%20compartments.%20In%20addition%2C%20the%20observation%20that%20cycling%20activity%20of%20stem%20cells%20is%20related%20to%20the%20maximal%20lifespan%20of%20the%20mouse%20strain%20may%20open%20ways%20to%20identify%20the%20genetic%20mechanisms%20of%20both%20strain-%20and%20age-dependent%20variation%20in%20the%20structure%20of%20primitive%20hematopoietic%20cell%20compartments.%22%2C%22date%22%3A%22Mar%2001%2C%201997%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%22%22%2C%22ISSN%22%3A%220006-4971%22%2C%22url%22%3A%22http%3A%5C%2F%5C%2Fwww.bloodjournal.org%5C%2Fcontent%5C%2F89%5C%2F5%5C%2F1543%5C%2Ftab-article-info%22%2C%22collections%22%3A%5B%22FXC3FTC4%22%5D%2C%22dateModified%22%3A%222019-06-27T01%3A54%3A27Z%22%7D%7D%2C%7B%22key%22%3A%228J8FRJPG%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Mitchell%20et%20al.%22%2C%22parsedDate%22%3A%222019-01-08%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EMitchell%20SJ%2C%20Bernier%20M%2C%20Mattison%20JA%2C%20et%20al%20%282019%29%20Daily%20Fasting%20Improves%20Health%20and%20Survival%20in%20Male%20Mice%20Independent%20of%20Diet%20Composition%20and%20Calories.%20Cell%20Metab%2029%3A221-228.e3.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cmet.2018.08.011%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cmet.2018.08.011%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Daily%20Fasting%20Improves%20Health%20and%20Survival%20in%20Male%20Mice%20Independent%20of%20Diet%20Composition%20and%20Calories%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sarah%20J.%22%2C%22lastName%22%3A%22Mitchell%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Michel%22%2C%22lastName%22%3A%22Bernier%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Julie%20A.%22%2C%22lastName%22%3A%22Mattison%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Miguel%20A.%22%2C%22lastName%22%3A%22Aon%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tamzin%20A.%22%2C%22lastName%22%3A%22Kaiser%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22R.%20Michael%22%2C%22lastName%22%3A%22Anson%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yuji%22%2C%22lastName%22%3A%22Ikeno%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Rozalyn%20M.%22%2C%22lastName%22%3A%22Anderson%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Donald%20K.%22%2C%22lastName%22%3A%22Ingram%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Rafael%22%2C%22lastName%22%3A%22de%20Cabo%22%7D%5D%2C%22abstractNote%22%3A%22The%20importance%20of%20dietary%20composition%20and%20feeding%20patterns%20in%20aging%20remains%20largely%20unexplored%2C%20but%20was%20implicated%20recently%20in%20two%20prominent%20nonhuman%20primate%20studies.%20Here%2C%20we%20directly%20compare%20in%20mice%20the%20two%20diets%20used%20in%20the%20primate%20studies%20focusing%20on%20three%20paradigms%3A%20ad%20libitum%20%28AL%29%2C%2030%25%20calorie%20restriction%20%28CR%29%2C%20and%20single-meal%20feeding%20%28MF%29%2C%20which%20accounts%20for%20differences%20in%20energy%20density%20and%20caloric%20intake%20consumed%20by%20the%20AL%20mice.%20MF%20and%20CR%20regimes%20enhanced%20longevity%20regardless%20of%20diet%20composition%2C%20which%20alone%20had%20no%20significant%20impact%20within%20feeding%20regimens.%20Like%20CR%20animals%2C%20MF%20mice%20ate%20quickly%2C%20imposing%20periods%20of%20extended%20daily%20fasting%20on%20themselves%20that%20produced%20significant%20improvements%20in%20morbidity%20and%20mortality%20compared%20with%20AL.%20These%20health%20and%20survival%20benefits%20conferred%20by%20periods%20of%20extended%20daily%20fasting%2C%20independent%20of%20dietary%20composition%2C%20have%20major%20implications%20for%20human%20health%20and%20clinical%20applicability.%22%2C%22date%22%3A%22Jan%2008%2C%202019%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.cmet.2018.08.011%22%2C%22ISSN%22%3A%221932-7420%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22UIM8YXRX%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A02%3A24Z%22%7D%7D%2C%7B%22key%22%3A%22J7ZVR9HH%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Hutchison%20et%20al.%22%2C%22parsedDate%22%3A%222019-05%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EHutchison%20AT%2C%20Regmi%20P%2C%20Manoogian%20ENC%2C%20et%20al%20%282019%29%20Time-Restricted%20Feeding%20Improves%20Glucose%20Tolerance%20in%20Men%20at%20Risk%20for%20Type%202%20Diabetes%3A%20A%20Randomized%20Crossover%20Trial.%20Obesity%20%28Silver%20Spring%29%2027%3A724%26%23x2013%3B732.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Foby.22449%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1002%5C%2Foby.22449%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Time-Restricted%20Feeding%20Improves%20Glucose%20Tolerance%20in%20Men%20at%20Risk%20for%20Type%202%20Diabetes%3A%20A%20Randomized%20Crossover%20Trial%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Amy%20T.%22%2C%22lastName%22%3A%22Hutchison%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Prashant%22%2C%22lastName%22%3A%22Regmi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Emily%20N.%20C.%22%2C%22lastName%22%3A%22Manoogian%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jason%20G.%22%2C%22lastName%22%3A%22Fleischer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gary%20A.%22%2C%22lastName%22%3A%22Wittert%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Satchidananda%22%2C%22lastName%22%3A%22Panda%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Leonie%20K.%22%2C%22lastName%22%3A%22Heilbronn%22%7D%5D%2C%22abstractNote%22%3A%22OBJECTIVE%3A%20This%20study%20aimed%20to%20assess%20the%20effects%20of%209-hour%20time-restricted%20feeding%20%28TRF%29%2C%20early%20%28TRFe%29%20or%20delayed%20%28TRFd%29%2C%20on%20glucose%20tolerance%20in%20men%20at%20risk%20for%20type%202%20diabetes.%5CnMETHODS%3A%20Fifteen%20men%20%28age%2055%5Cu00a0%5Cu00b1%5Cu00a03%20years%2C%20BMI%2033.9%5Cu00a0%5Cu00b1%5Cu00a00.8%5Cu00a0kg%5C%2Fm2%20%29%20wore%20a%20continuous%20glucose%20monitor%20for%207%5Cu00a0days%20of%20baseline%20assessment%20and%20during%20two%207-day%20TRF%20conditions.%20Participants%20were%20randomized%20to%20TRFe%20%288%5Cu00a0am%20to%205%20pm%29%20or%20TRFd%20%2812%20pm%20to%209%20pm%29%2C%20separated%20by%20a%202-week%20washout%20phase.%20Glucose%2C%20insulin%2C%20triglycerides%2C%20nonesterified%20fatty%20acids%2C%20and%20gastrointestinal%20hormone%20incremental%20areas%20under%20the%20curve%20were%20calculated%20following%20a%20standard%20meal%20on%20days%200%20and%207%20at%208%5Cu00a0am%20%28TRFe%29%20or%2012%20pm%20%28TRFd%29.%5CnRESULTS%3A%20TRF%20improved%20glucose%20tolerance%20as%20assessed%20by%20a%20reduction%20in%20glucose%20incremental%20area%20under%20the%20curve%20%28P%5Cu00a0%3D%5Cu00a00.001%29%20and%20fasting%20triglycerides%20%28P%5Cu00a0%3D%5Cu00a00.003%29%20on%20day%207%20versus%20day%200.%20However%2C%20there%20were%20no%20mealtime%20by%20TRF%20interactions%20in%20any%20of%20the%20variables%20examined.%20There%20was%20also%20no%20effect%20of%20TRF%20on%20fasting%20and%20postprandial%20insulin%2C%20nonesterified%20fatty%20acids%2C%20or%20gastrointestinal%20hormones.%20Mean%20fasting%20glucose%20by%20continuous%20glucose%20monitor%20was%20lower%20in%20TRFe%20%28P%5Cu00a0%3D%5Cu00a00.02%29%20but%20not%20TRFd%20%28P%5Cu00a0%3D%5Cu00a00.17%29%20versus%20baseline%2C%20but%20there%20was%20no%20difference%20between%20TRF%20conditions.%5CnCONCLUSIONS%3A%20While%20only%20TRFe%20lowered%20mean%20fasting%20glucose%2C%20TRF%20improved%20glycemic%20responses%20to%20a%20test%20meal%20in%20men%20at%20risk%20for%20type%202%20diabetes%20regardless%20of%20the%20clock%20time%20that%20TRF%20was%20initiated.%22%2C%22date%22%3A%22May%202019%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1002%5C%2Foby.22449%22%2C%22ISSN%22%3A%221930-739X%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22UIM8YXRX%22%2C%226BPRM5LB%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A02%3A23Z%22%7D%7D%2C%7B%22key%22%3A%22SQU9ZGIQ%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Wei%20et%20al.%22%2C%22parsedDate%22%3A%222017-02-15%22%2C%22numChildren%22%3A3%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EWei%20M%2C%20Brandhorst%20S%2C%20Shelehchi%20M%2C%20et%20al%20%282017%29%20Fasting-mimicking%20diet%20and%20markers%5C%2Frisk%20factors%20for%20aging%2C%20diabetes%2C%20cancer%2C%20and%20cardiovascular%20disease.%20Science%20Translational%20Medicine%209%3Aeaai8700.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1126%5C%2Fscitranslmed.aai8700%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1126%5C%2Fscitranslmed.aai8700%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Fasting-mimicking%20diet%20and%20markers%5C%2Frisk%20factors%20for%20aging%2C%20diabetes%2C%20cancer%2C%20and%20cardiovascular%20disease%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Min%22%2C%22lastName%22%3A%22Wei%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastian%22%2C%22lastName%22%3A%22Brandhorst%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mahshid%22%2C%22lastName%22%3A%22Shelehchi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hamed%22%2C%22lastName%22%3A%22Mirzaei%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Chia%20Wei%22%2C%22lastName%22%3A%22Cheng%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Julia%22%2C%22lastName%22%3A%22Budniak%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Susan%22%2C%22lastName%22%3A%22Groshen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Wendy%20J.%22%2C%22lastName%22%3A%22Mack%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Esra%22%2C%22lastName%22%3A%22Guen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Stefano%20Di%22%2C%22lastName%22%3A%22Biase%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pinchas%22%2C%22lastName%22%3A%22Cohen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Todd%20E.%22%2C%22lastName%22%3A%22Morgan%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tanya%22%2C%22lastName%22%3A%22Dorff%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kurt%22%2C%22lastName%22%3A%22Hong%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Andreas%22%2C%22lastName%22%3A%22Michalsen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Alessandro%22%2C%22lastName%22%3A%22Laviano%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Valter%20D.%22%2C%22lastName%22%3A%22Longo%22%7D%5D%2C%22abstractNote%22%3A%22Fasting%3A%20More%20than%20a%20fad%5CnMice%20that%20fast%20periodically%20are%20healthier%2C%20metabolically%20speaking.%20To%20explore%20whether%20fasting%20can%20help%20people%20as%20well%2C%20Wei%20et%20al.%20studied%2071%20people%20who%20either%20consumed%20a%20fasting-mimicking%20diet%20for%205%20days%20each%20month%20for%203%20months%20or%20maintained%20their%20normal%20diet%20for%203%20months%20and%20then%20switched%20to%20the%20fasting%20schedule.%20The%20fasting-like%20diet%20reduced%20body%20weight%20and%20body%20fat%2C%20lowered%20blood%20pressure%2C%20and%20decreased%20the%20hormone%20IGF-1%2C%20which%20has%20been%20implicated%20in%20aging%20and%20disease.%20A%20post%20hoc%20analysis%20replicated%20these%20results%20and%20also%20showed%20that%20fasting%20decreased%20BMI%2C%20glucose%2C%20triglycerides%2C%20cholesterol%2C%20and%20C-reactive%20protein%20%28a%20marker%20for%20inflammation%29.%20These%20effects%20were%20generally%20larger%20in%20the%20subjects%20who%20were%20at%20greater%20risk%20of%20disease%20at%20the%20start%20of%20the%20study.%20A%20larger%20study%20is%20needed%20to%20replicate%20these%20results%2C%20but%20they%20raise%20the%20possibility%20that%20fasting%20may%20be%20a%20practical%20road%20to%20a%20healthy%20metabolic%20system.%5CnA%20periodic%20fasting%20diet%20improves%20markers%20of%20metabolic%20dysfunction%2C%20particularly%20in%20people%20already%20at%20risk.%5CnA%20periodic%20fasting%20diet%20improves%20markers%20of%20metabolic%20dysfunction%2C%20particularly%20in%20people%20already%20at%20risk.%22%2C%22date%22%3A%222017%5C%2F02%5C%2F15%22%2C%22language%22%3A%22en%22%2C%22DOI%22%3A%2210.1126%5C%2Fscitranslmed.aai8700%22%2C%22ISSN%22%3A%221946-6234%2C%201946-6242%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fstm.sciencemag.org%5C%2Fcontent%5C%2F9%5C%2F377%5C%2Feaai8700%22%2C%22collections%22%3A%5B%22UXEBAQIK%22%2C%22XPHE39C6%22%2C%22JQMRJMTS%22%2C%22UZR7UXN9%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A02%3A20Z%22%7D%7D%2C%7B%22key%22%3A%229N4I95G9%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Newman%20et%20al.%22%2C%22parsedDate%22%3A%222017-09-05%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ENewman%20JC%2C%20Covarrubias%20AJ%2C%20Zhao%20M%2C%20et%20al%20%282017%29%20Ketogenic%20Diet%20Reduces%20Midlife%20Mortality%20and%20Improves%20Memory%20in%20Aging%20Mice.%20Cell%20Metab%2026%3A547-557.e8.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cmet.2017.08.004%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cmet.2017.08.004%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Ketogenic%20Diet%20Reduces%20Midlife%20Mortality%20and%20Improves%20Memory%20in%20Aging%20Mice%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22John%20C.%22%2C%22lastName%22%3A%22Newman%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Anthony%20J.%22%2C%22lastName%22%3A%22Covarrubias%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Minghao%22%2C%22lastName%22%3A%22Zhao%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Xinxing%22%2C%22lastName%22%3A%22Yu%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Philipp%22%2C%22lastName%22%3A%22Gut%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Che-Ping%22%2C%22lastName%22%3A%22Ng%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yu%22%2C%22lastName%22%3A%22Huang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Saptarsi%22%2C%22lastName%22%3A%22Haldar%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eric%22%2C%22lastName%22%3A%22Verdin%22%7D%5D%2C%22abstractNote%22%3A%22Ketogenic%20diets%20recapitulate%20certain%20metabolic%20aspects%20of%20dietary%20restriction%20such%20as%20reliance%20on%20fatty%5Cu00a0acid%20metabolism%20and%20production%20of%20ketone%20bodies.%20We%20investigated%20whether%20an%20isoprotein%20ketogenic%20diet%20%28KD%29%20might%2C%20like%20dietary%20restriction%2C%20affect%20longevity%20and%20healthspan%20in%20C57BL%5C%2F6%20male%20mice.%20We%20find%20that%20Cyclic%20KD%2C%20KD%20alternated%20weekly%20with%20the%20Control%20diet%20to%20prevent%20obesity%2C%20reduces%20midlife%20mortality%20but%20does%20not%20affect%20maximum%20lifespan.%20A%20non-ketogenic%20high-fat%20diet%20%28HF%29%20fed%20similarly%20may%20have%20an%20intermediate%20effect%20on%20mortality.%20Cyclic%20KD%20improves%20memory%20performance%20in%20old%20age%2C%20while%20modestly%20improving%20composite%20healthspan%20measures.%20Gene%20expression%20analysis%20identifies%20downregulation%20of%20insulin%2C%20protein%20synthesis%2C%20and%20fatty%20acid%20synthesis%20pathways%20as%20mechanisms%20common%20to%20KD%20and%20HF.%20However%2C%20upregulation%20of%20PPAR%5Cu03b1%20target%20genes%20is%20unique%20to%20KD%2C%20consistent%20across%20tissues%2C%20and%20preserved%20in%20old%20age.%20In%20all%2C%20we%20show%20that%20a%20non-obesogenic%20ketogenic%20diet%20improves%20survival%2C%20memory%2C%20and%20healthspan%20in%20aging%20mice.%22%2C%22date%22%3A%22Sep%2005%2C%202017%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.cmet.2017.08.004%22%2C%22ISSN%22%3A%221932-7420%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22IB4QNEHC%22%2C%22BEIYKDRT%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A02%3A17Z%22%7D%7D%2C%7B%22key%22%3A%22CXS8BFWR%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Gill%20and%20Panda%22%2C%22parsedDate%22%3A%222015-11-03%22%2C%22numChildren%22%3A4%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EGill%20S%2C%20Panda%20S%20%282015%29%20A%20Smartphone%20App%20Reveals%20Erratic%20Diurnal%20Eating%20Patterns%20in%20Humans%20that%20Can%20Be%20Modulated%20for%20Health%20Benefits.%20Cell%20Metab%2022%3A789%26%23x2013%3B798.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cmet.2015.09.005%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cmet.2015.09.005%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22A%20Smartphone%20App%20Reveals%20Erratic%20Diurnal%20Eating%20Patterns%20in%20Humans%20that%20Can%20Be%20Modulated%20for%20Health%20Benefits%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Shubhroz%22%2C%22lastName%22%3A%22Gill%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Satchidananda%22%2C%22lastName%22%3A%22Panda%22%7D%5D%2C%22abstractNote%22%3A%22A%20diurnal%20rhythm%20of%20eating-fasting%20promotes%20health%2C%20but%20the%20eating%20pattern%20of%20humans%20is%20rarely%20assessed.%20Using%20a%20mobile%20app%2C%20we%20monitored%20ingestion%20events%20in%20healthy%20adults%20with%20no%20shift-work%20for%20several%20days.%20Most%20subjects%20ate%20frequently%20and%20erratically%20throughout%20wakeful%20hours%2C%20and%20overnight%20fasting%20duration%20paralleled%20time%20in%20bed.%20There%20was%20a%20bias%20toward%20eating%20late%2C%20with%20an%20estimated%20%3C25%25%20of%20calories%20being%20consumed%20before%20noon%20and%20%3E35%25%20after%206%20p.m.%20%5C%22Metabolic%20jetlag%5C%22%20resulting%20from%20weekday%5C%2Fweekend%20variation%20in%20eating%20pattern%20akin%20to%20travel%20across%20time%20zones%20was%20prevalent.%20The%20daily%20intake%20duration%20%2895%25%20interval%29%20exceeded%2014.75%5Cu00a0hr%20for%20half%20of%20the%20cohort.%20When%20overweight%20individuals%20with%20%3E14%5Cu00a0hr%20eating%20duration%20ate%20for%20only%2010-11%5Cu00a0hr%20daily%20for%2016%5Cu00a0weeks%20assisted%20by%20a%20data%20visualization%20%28raster%20plot%20of%20dietary%20intake%20pattern%2C%20%5C%22feedogram%5C%22%29%20that%20we%5Cu00a0developed%2C%20they%20reduced%20body%20weight%2C%20reported%20being%20energetic%2C%20and%20improved%20sleep.%20Benefits%20persisted%20for%20a%20year.%22%2C%22date%22%3A%22Nov%2003%2C%202015%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.cmet.2015.09.005%22%2C%22ISSN%22%3A%221932-7420%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22UIM8YXRX%22%2C%226BPRM5LB%22%2C%227V6YMPXS%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A02%3A12Z%22%7D%7D%2C%7B%22key%22%3A%223HHU6VZN%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Brandhorst%20et%20al.%22%2C%22parsedDate%22%3A%222015-07-07%22%2C%22numChildren%22%3A3%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EBrandhorst%20S%2C%20Choi%20IY%2C%20Wei%20M%2C%20et%20al%20%282015%29%20A%20Periodic%20Diet%20that%20Mimics%20Fasting%20Promotes%20Multi-System%20Regeneration%2C%20Enhanced%20Cognitive%20Performance%2C%20and%20Healthspan.%20Cell%20Metab%2022%3A86%26%23x2013%3B99.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cmet.2015.05.012%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cmet.2015.05.012%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22A%20Periodic%20Diet%20that%20Mimics%20Fasting%20Promotes%20Multi-System%20Regeneration%2C%20Enhanced%20Cognitive%20Performance%2C%20and%20Healthspan%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastian%22%2C%22lastName%22%3A%22Brandhorst%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22In%20Young%22%2C%22lastName%22%3A%22Choi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Min%22%2C%22lastName%22%3A%22Wei%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Chia%20Wei%22%2C%22lastName%22%3A%22Cheng%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sargis%22%2C%22lastName%22%3A%22Sedrakyan%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gerardo%22%2C%22lastName%22%3A%22Navarrete%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Louis%22%2C%22lastName%22%3A%22Dubeau%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Li%20Peng%22%2C%22lastName%22%3A%22Yap%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ryan%22%2C%22lastName%22%3A%22Park%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Manlio%22%2C%22lastName%22%3A%22Vinciguerra%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Stefano%22%2C%22lastName%22%3A%22Di%20Biase%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hamed%22%2C%22lastName%22%3A%22Mirzaei%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mario%20G.%22%2C%22lastName%22%3A%22Mirisola%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Patra%22%2C%22lastName%22%3A%22Childress%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Lingyun%22%2C%22lastName%22%3A%22Ji%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Susan%22%2C%22lastName%22%3A%22Groshen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fabio%22%2C%22lastName%22%3A%22Penna%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Patrizio%22%2C%22lastName%22%3A%22Odetti%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Laura%22%2C%22lastName%22%3A%22Perin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Peter%20S.%22%2C%22lastName%22%3A%22Conti%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yuji%22%2C%22lastName%22%3A%22Ikeno%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Brian%20K.%22%2C%22lastName%22%3A%22Kennedy%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pinchas%22%2C%22lastName%22%3A%22Cohen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Todd%20E.%22%2C%22lastName%22%3A%22Morgan%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tanya%20B.%22%2C%22lastName%22%3A%22Dorff%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Valter%20D.%22%2C%22lastName%22%3A%22Longo%22%7D%5D%2C%22abstractNote%22%3A%22Prolonged%20fasting%20%28PF%29%20promotes%20stress%20resistance%2C%20but%20its%20effects%20on%20longevity%20are%20poorly%20understood.%20We%20show%20that%20alternating%20PF%20and%20nutrient-rich%20medium%20extended%20yeast%20lifespan%20independently%20of%20established%20pro-longevity%20genes.%20In%20mice%2C%204%20days%20of%20a%20diet%20that%20mimics%20fasting%20%28FMD%29%2C%20developed%20to%20minimize%20the%20burden%20of%20PF%2C%20decreased%20the%20size%20of%20multiple%20organs%5C%2Fsystems%2C%20an%20effect%20followed%20upon%20re-feeding%20by%20an%20elevated%20number%20of%20progenitor%20and%20stem%20cells%20and%20regeneration.%20Bi-monthly%20FMD%20cycles%20started%20at%20middle%20age%20extended%20longevity%2C%20lowered%20visceral%20fat%2C%20reduced%20cancer%20incidence%20and%20skin%20lesions%2C%20rejuvenated%20the%20immune%20system%2C%20and%20retarded%20bone%20mineral%20density%20loss.%20In%20old%20mice%2C%20FMD%20cycles%20promoted%20hippocampal%20neurogenesis%2C%20lowered%20IGF-1%20levels%20and%20PKA%20activity%2C%20elevated%20NeuroD1%2C%20and%20improved%20cognitive%20performance.%20In%20a%20pilot%20clinical%20trial%2C%20three%20FMD%20cycles%20decreased%20risk%20factors%5C%2Fbiomarkers%20for%20aging%2C%20diabetes%2C%20cardiovascular%20disease%2C%20and%20cancer%20without%20major%20adverse%20effects%2C%20providing%20support%20for%20the%20use%20of%20FMDs%20to%20promote%20healthspan.%22%2C%22date%22%3A%22Jul%2007%2C%202015%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.cmet.2015.05.012%22%2C%22ISSN%22%3A%221932-7420%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22UXEBAQIK%22%2C%22GZ6NNKTA%22%2C%22NZZRKCN6%22%2C%22XPHE39C6%22%2C%22UZR7UXN9%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A02%3A11Z%22%7D%7D%2C%7B%22key%22%3A%223WVQ9RWW%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Cheng%20et%20al.%22%2C%22parsedDate%22%3A%222014-06-05%22%2C%22numChildren%22%3A3%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ECheng%20C-W%2C%20Adams%20GB%2C%20Perin%20L%2C%20et%20al%20%282014%29%20Prolonged%20fasting%20reduces%20IGF-1%5C%2FPKA%20to%20promote%20hematopoietic-stem-cell-based%20regeneration%20and%20reverse%20immunosuppression.%20Cell%20Stem%20Cell%2014%3A810%26%23x2013%3B823.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.stem.2014.04.014%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.stem.2014.04.014%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Prolonged%20fasting%20reduces%20IGF-1%5C%2FPKA%20to%20promote%20hematopoietic-stem-cell-based%20regeneration%20and%20reverse%20immunosuppression%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Chia-Wei%22%2C%22lastName%22%3A%22Cheng%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gregor%20B.%22%2C%22lastName%22%3A%22Adams%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Laura%22%2C%22lastName%22%3A%22Perin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Min%22%2C%22lastName%22%3A%22Wei%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Xiaoying%22%2C%22lastName%22%3A%22Zhou%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ben%20S.%22%2C%22lastName%22%3A%22Lam%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Stefano%22%2C%22lastName%22%3A%22Da%20Sacco%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mario%22%2C%22lastName%22%3A%22Mirisola%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22David%20I.%22%2C%22lastName%22%3A%22Quinn%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tanya%20B.%22%2C%22lastName%22%3A%22Dorff%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22John%20J.%22%2C%22lastName%22%3A%22Kopchick%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Valter%20D.%22%2C%22lastName%22%3A%22Longo%22%7D%5D%2C%22abstractNote%22%3A%22Immune%20system%20defects%20are%20at%20the%20center%20of%20aging%20and%20a%20range%20of%20diseases.%20Here%2C%20we%20show%20that%20prolonged%20fasting%20reduces%20circulating%20IGF-1%20levels%20and%20PKA%20activity%20in%20various%20cell%20populations%2C%20leading%20to%20signal%20transduction%20changes%20in%20long-term%20hematopoietic%20stem%20cells%20%28LT-HSCs%29%20and%20niche%20cells%20that%20promote%20stress%20resistance%2C%20self-renewal%2C%20and%20lineage-balanced%20regeneration.%20Multiple%20cycles%20of%20fasting%20abated%20the%20immunosuppression%20and%20mortality%20caused%20by%20chemotherapy%20and%20reversed%20age-dependent%20myeloid-bias%20in%20mice%2C%20in%20agreement%20with%20preliminary%20data%20on%20the%20protection%20of%20lymphocytes%20from%20chemotoxicity%20in%20fasting%20patients.%20The%20proregenerative%20effects%20of%20fasting%20on%20stem%20cells%20were%20recapitulated%20by%20deficiencies%20in%20either%20IGF-1%20or%20PKA%20and%20blunted%20by%20exogenous%20IGF-1.%20These%20findings%20link%20the%20reduced%20levels%20of%20IGF-1%20caused%20by%20fasting%20to%20PKA%20signaling%20and%20establish%20their%20crucial%20role%20in%20regulating%20hematopoietic%20stem%20cell%20protection%2C%20self-renewal%2C%20and%20regeneration.%22%2C%22date%22%3A%22Jun%2005%2C%202014%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.stem.2014.04.014%22%2C%22ISSN%22%3A%221875-9777%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22KMY5B6AS%22%2C%2247PY442C%22%2C%22GZ6NNKTA%22%2C%22NZZRKCN6%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A02%3A09Z%22%7D%7D%2C%7B%22key%22%3A%22P4DNECM5%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Naito%20et%20al.%22%2C%22parsedDate%22%3A%222013-07-19%22%2C%22numChildren%22%3A3%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ENaito%20T%2C%20Kuma%20A%2C%20Mizushima%20N%20%282013%29%20Differential%20contribution%20of%20insulin%20and%20amino%20acids%20to%20the%20mTORC1-autophagy%20pathway%20in%20the%20liver%20and%20muscle.%20J%20Biol%20Chem%20288%3A21074%26%23x2013%3B21081.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1074%5C%2Fjbc.M113.456228%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1074%5C%2Fjbc.M113.456228%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Differential%20contribution%20of%20insulin%20and%20amino%20acids%20to%20the%20mTORC1-autophagy%20pathway%20in%20the%20liver%20and%20muscle%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takako%22%2C%22lastName%22%3A%22Naito%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Akiko%22%2C%22lastName%22%3A%22Kuma%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Noboru%22%2C%22lastName%22%3A%22Mizushima%22%7D%5D%2C%22abstractNote%22%3A%22Autophagy%20is%20a%20highly%20inducible%20intracellular%20degradation%20process.%20It%20is%20generally%20induced%20by%20nutrient%20starvation%20and%20suppressed%20by%20food%20intake.%20Mammalian%20%28or%20mechanistic%29%20target%20of%20rapamycin%20complex%201%20%28mTORC1%29%20is%20considered%20to%20be%20the%20major%20regulator%20of%20autophagy%2C%20but%20the%20precise%20mechanism%20of%20in%20vivo%20regulation%20remains%20to%20be%20fully%20characterized.%20Here%2C%20we%20examined%20the%20autophagy-suppressive%20effect%20of%20glucose%2C%20insulin%2C%20and%20amino%20acids%20in%20the%20liver%20and%20muscle%20in%20mice%20starved%20for%201%20day.%20Refeeding%20after%20starvation%20with%20a%20standard%20mouse%20chow%20rapidly%20suppressed%20autophagy%20in%20both%20tissues%2C%20and%20this%20suppression%20was%20inhibited%20by%20rapamycin%20administration%20almost%20completely%20in%20the%20liver%20and%20partially%20in%20muscle%2C%20confirming%20that%20mTORC1%20is%20indeed%20a%20crucial%20regulator%20in%20vivo.%20As%20glucose%20administration%20showed%20no%20major%20suppressive%20effect%20on%20autophagy%2C%20we%20examined%20the%20role%20of%20insulin%20and%20amino%20acids%20using%20hyperinsulinemic-euglycemic%20clamp%20and%20intravenous%20amino%20acid%20infusion%20techniques.%20Insulin%20administration%20showed%20a%20clear%20effect%20on%20the%20mTORC1-autophagy%20pathway%20in%20muscle%2C%20but%20had%20only%20a%20very%20weak%20effect%20in%20the%20liver.%20By%20contrast%2C%20amino%20acids%20were%20able%20to%20regulate%20the%20mTORC1-autophagy%20pathway%20in%20the%20liver%2C%20but%20less%20effectively%20in%20muscle.%20These%20results%20suggest%20that%20autophagy%20is%20differentially%20regulated%20by%20insulin%20and%20amino%20acids%20in%20a%20tissue-dependent%20manner.%22%2C%22date%22%3A%22Jul%2019%2C%202013%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1074%5C%2Fjbc.M113.456228%22%2C%22ISSN%22%3A%221083-351X%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22FM3XX4QZ%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A02%3A08Z%22%7D%7D%2C%7B%22key%22%3A%229R8B7N39%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Browning%20et%20al.%22%2C%22parsedDate%22%3A%222012-03%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EBrowning%20JD%2C%20Baxter%20J%2C%20Satapati%20S%2C%20Burgess%20SC%20%282012%29%20The%20effect%20of%20short-term%20fasting%20on%20liver%20and%20skeletal%20muscle%20lipid%2C%20glucose%2C%20and%20energy%20metabolism%20in%20healthy%20women%20and%20men.%20J%20Lipid%20Res%2053%3A577%26%23x2013%3B586.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1194%5C%2Fjlr.P020867%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1194%5C%2Fjlr.P020867%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22The%20effect%20of%20short-term%20fasting%20on%20liver%20and%20skeletal%20muscle%20lipid%2C%20glucose%2C%20and%20energy%20metabolism%20in%20healthy%20women%20and%20men%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jeffrey%20D.%22%2C%22lastName%22%3A%22Browning%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jeannie%22%2C%22lastName%22%3A%22Baxter%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Santhosh%22%2C%22lastName%22%3A%22Satapati%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Shawn%20C.%22%2C%22lastName%22%3A%22Burgess%22%7D%5D%2C%22abstractNote%22%3A%22Fasting%20promotes%20triglyceride%20%28TG%29%20accumulation%20in%20lean%20tissues%20of%20some%20animals%2C%20but%20the%20effect%20in%20humans%20is%20unknown.%20Additionally%2C%20fasting%20lipolysis%20is%20sexually%20dimorphic%20in%20humans%2C%20suggesting%20that%20lean%20tissue%20TG%20accumulation%20and%20metabolism%20may%20differ%20between%20women%20and%20men.%20This%20study%20investigated%20lean%20tissue%20TG%20content%20and%20metabolism%20in%20women%20and%20men%20during%20extended%20fasting.%20Liver%20and%20muscle%20TG%20content%20were%20measured%20by%20magnetic%20resonance%20spectroscopy%20during%20a%2048-h%20fast%20in%20healthy%20men%20and%20women.%20Whole-body%20and%20hepatic%20carbohydrate%2C%20lipid%2C%20and%20energy%20metabolism%20were%20also%20evaluated%20using%20biochemical%2C%20calorimetric%2C%20and%20stable%20isotope%20tracer%20techniques.%20As%20expected%2C%20postabsorptive%20plasma%20fatty%20acids%20%28FAs%29%20were%20higher%20in%20women%20than%20in%20men%20but%20increased%20more%20rapidly%20in%20men%20with%20the%20onset%20of%20early%20starvation.%20Concurrently%2C%20sexual%20dimorphism%20was%20apparent%20in%20lean%20tissue%20TG%20accumulation%20during%20the%20fast%2C%20occurring%20in%20livers%20of%20men%20but%20in%20muscles%20of%20women.%20Despite%20differences%20in%20lean%20tissue%20TG%20distribution%2C%20men%20and%20women%20had%20identical%20fasting%20responses%20in%20whole-body%20and%20hepatic%20glucose%20and%20oxidative%20metabolism.%20In%20conclusion%2C%20TG%20accumulated%20in%20livers%20of%20men%20but%20in%20muscles%20of%20women%20during%20extended%20fasting.%20This%20sexual%20dimorphism%20was%20related%20to%20differential%20fasting%20plasma%20FA%20concentrations%20but%20not%20to%20whole%20body%20or%20hepatic%20utilization%20of%20this%20substrate.%22%2C%22date%22%3A%222012-3%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%2210.1194%5C%2Fjlr.P020867%22%2C%22ISSN%22%3A%220022-2275%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fwww.ncbi.nlm.nih.gov%5C%2Fpmc%5C%2Farticles%5C%2FPMC3276482%5C%2F%22%2C%22collections%22%3A%5B%2247PY442C%22%2C%22565CW9N6%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A02%3A06Z%22%7D%7D%2C%7B%22key%22%3A%22338PM6JJ%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Lee%20et%20al.%22%2C%22parsedDate%22%3A%222010-02-15%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ELee%20C%2C%20Safdie%20FM%2C%20Raffaghello%20L%2C%20et%20al%20%282010%29%20Reduced%20levels%20of%20IGF-I%20mediate%20differential%20protection%20of%20normal%20and%20cancer%20cells%20in%20response%20to%20fasting%20and%20improve%20chemotherapeutic%20index.%20Cancer%20Res%2070%3A1564%26%23x2013%3B1572.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1158%5C%2F0008-5472.CAN-09-3228%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1158%5C%2F0008-5472.CAN-09-3228%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Reduced%20levels%20of%20IGF-I%20mediate%20differential%20protection%20of%20normal%20and%20cancer%20cells%20in%20response%20to%20fasting%20and%20improve%20chemotherapeutic%20index%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Changhan%22%2C%22lastName%22%3A%22Lee%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fernando%20M.%22%2C%22lastName%22%3A%22Safdie%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Lizzia%22%2C%22lastName%22%3A%22Raffaghello%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Min%22%2C%22lastName%22%3A%22Wei%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Federica%22%2C%22lastName%22%3A%22Madia%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Edoardo%22%2C%22lastName%22%3A%22Parrella%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22David%22%2C%22lastName%22%3A%22Hwang%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pinchas%22%2C%22lastName%22%3A%22Cohen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Giovanna%22%2C%22lastName%22%3A%22Bianchi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Valter%20D.%22%2C%22lastName%22%3A%22Longo%22%7D%5D%2C%22abstractNote%22%3A%22Inhibitors%20of%20the%20insulin-like%20growth%20factor-I%20%28IGF-I%29%20receptor%20have%20been%20widely%20studied%20for%20their%20ability%20to%20enhance%20the%20killing%20of%20a%20variety%20of%20malignant%20cells%2C%20but%20whether%20IGF-I%20signaling%20differentially%20protects%20the%20host%20and%20cancer%20cells%20against%20chemotherapy%20is%20unknown.%20Starvation%20can%20protect%20mice%2C%20but%20not%20cancer%20cells%2C%20against%20high-dose%20chemotherapy%20%5Bdifferential%20stress%20resistance%20%28DSR%29%5D.%20Here%2C%20we%20offer%20evidence%20that%20IGF-I%20reduction%20mediates%20part%20of%20the%20starvation-dependent%20DSR.%20A%2072-hour%20fast%20in%20mice%20reduced%20circulating%20IGF-I%20by%2070%25%20and%20increased%20the%20level%20of%20the%20IGF-I%20inhibitor%20IGFBP-1%20by%2011-fold.%20LID%20mice%2C%20with%20a%2070%25%20to%2080%25%20reduction%20in%20circulating%20IGF-I%20levels%2C%20were%20protected%20against%20three%20of%20four%20chemotherapy%20drugs%20tested.%20Restoration%20of%20IGF-I%20was%20sufficient%20to%20reverse%20the%20protective%20effect%20of%20fasting.%20Sixty%20percent%20of%20melanoma-bearing%20LID%20mice%20treated%20with%20doxorubicin%20achieved%20long-term%20survival%20whereas%20all%20control%20mice%20died%20of%20either%20metastases%20or%20chemotherapy%20toxicity.%20Reducing%20IGF-I%5C%2FIGF-I%20signaling%20protected%20primary%20glia%2C%20but%20not%20glioma%20cells%2C%20against%20cyclophosphamide%20and%20protected%20mouse%20embryonic%20fibroblasts%20against%20doxorubicin.%20Further%2C%20S.%20cerevisiae%20lacking%20homologs%20of%20IGF-I%20signaling%20proteins%20were%20protected%20against%20chemotherapy-dependent%20DNA%20damage%20in%20a%20manner%20that%20could%20be%20reversed%20by%20expressing%20a%20constitutively%20active%20form%20of%20Ras.%20We%20conclude%20that%20normal%20cells%20and%20mice%20can%20be%20protected%20against%20chemotherapy-dependent%20damage%20by%20reducing%20circulating%20IGF-I%20levels%20and%20by%20a%20mechanism%20that%20involves%20downregulation%20of%20proto-oncogene%20signals.%22%2C%22date%22%3A%22Feb%2015%2C%202010%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1158%5C%2F0008-5472.CAN-09-3228%22%2C%22ISSN%22%3A%221538-7445%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22KMY5B6AS%22%2C%22G537RKA2%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A02%3A05Z%22%7D%7D%2C%7B%22key%22%3A%224TLPENTP%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Marliss%20et%20al.%22%2C%22parsedDate%22%3A%221970-12%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EMarliss%20EB%2C%20Aoki%20TT%2C%20Unger%20RH%2C%20et%20al%20%281970%29%20Glucagon%20levels%20and%20metabolic%20effects%20in%20fasting%20man.%20J%20Clin%20Invest%2049%3A2256%26%23x2013%3B2270.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1172%5C%2FJCI106445%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1172%5C%2FJCI106445%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Glucagon%20levels%20and%20metabolic%20effects%20in%20fasting%20man%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22E.%20B.%22%2C%22lastName%22%3A%22Marliss%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22T.%20T.%22%2C%22lastName%22%3A%22Aoki%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22R.%20H.%22%2C%22lastName%22%3A%22Unger%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22J.%20S.%22%2C%22lastName%22%3A%22Soeldner%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22G.%20F.%22%2C%22lastName%22%3A%22Cahill%22%7D%5D%2C%22abstractNote%22%3A%22The%20role%20of%20glucagon%20in%20the%20metabolic%20adaptation%20to%20prolonged%20fasting%20in%20man%20has%20been%20examined.%20Plasma%20immunoreactive%20glucagon%20was%20determined%20during%206-wk%20fasts%20and%20during%20infusion%20of%20exogenous%20glucagon%20using%20an%20assay%20which%20minimized%20nonpancreatic%20immunoreactivity.%20Plasma%20glucagon%20concentrations%20rose%20twofold%20to%20a%20peak%20on%20the%203rd%20day%20of%20fasting%20and%20then%20declined%20thereafter%20to%20a%20level%20maintained%20at%20or%20above%20postabsorptive.%20Insulin%20concentration%20declined%20to%20a%20plateau%20by%20the%203rd%20day.%20Thus%20a%20persisting%20altered%20relationship%20of%20glucagon%20and%20insulin%20concentrations%20characterized%20the%20fasted%20state.%20A%20synergism%20of%20low%20insulin%20and%20relative%20or%20absolute%20elevation%20of%20glucagon%20levels%20is%20viewed%20as%20a%20hormonal%20mechanism%20controlling%20the%20rate%20of%20hepatic%20substrate%20extraction%20for%20gluconeogenesis.%20Glucagon%20was%20infused%20systemically%20into%204-6%20wk%20fasted%20subjects%20at%20three%20dose%20levels.%20A%20marked%20sensitivity%20of%20individual%20plasma%20free%20amino%20acids%20to%20the%20induced%20elevations%20of%20plasma%20glucagon%20within%20the%20physiologic%20range%20was%20demonstrated.%20At%20higher%20concentrations%2C%20equivalent%20to%20those%20present%20in%20the%20portal%20vein%2C%20stimulation%20of%20hepatic%20gluconeogenesis%20occurred%2C%20and%20the%20effects%20on%20glucose%2C%20insulin%2C%20and%20growth%20hormone%20levels%20and%20on%20ketone%20metabolism%20were%20induced.%22%2C%22date%22%3A%22Dec%201970%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1172%5C%2FJCI106445%22%2C%22ISSN%22%3A%220021-9738%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22KMY5B6AS%22%2C%22N5P5HQWF%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A01%3A57Z%22%7D%7D%2C%7B%22key%22%3A%22WX6XVYQC%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Owen%20et%20al.%22%2C%22parsedDate%22%3A%221969-03%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EOwen%20OE%2C%20Felig%20P%2C%20Morgan%20AP%2C%20et%20al%20%281969%29%20Liver%20and%20kidney%20metabolism%20during%20prolonged%20starvation.%20J%20Clin%20Invest%2048%3A574%26%23x2013%3B583.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1172%5C%2FJCI106016%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1172%5C%2FJCI106016%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Liver%20and%20kidney%20metabolism%20during%20prolonged%20starvation%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22O.%20E.%22%2C%22lastName%22%3A%22Owen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22P.%22%2C%22lastName%22%3A%22Felig%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%20P.%22%2C%22lastName%22%3A%22Morgan%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22J.%22%2C%22lastName%22%3A%22Wahren%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22G.%20F.%22%2C%22lastName%22%3A%22Cahill%22%7D%5D%2C%22abstractNote%22%3A%22This%20study%20quantifies%20the%20concentrations%20of%20circulating%20insulin%2C%20growth%20hormone%2C%20glucose%2C%20free%20fatty%20acids%2C%20glycerol%2C%20beta-hydroxybutyrate%2C%20acetoacetate%2C%20and%20alpha%20amino%20nitrogen%20in%2011%20obese%20subjects%20during%20prolonged%20starvation.%20The%20sites%20and%20estimated%20rates%20of%20gluconeogenesis%20and%20ketogenesis%20after%205-6%20wk%20of%20fasting%20were%20investigated%20in%20five%20of%20the%20subjects.%20Blood%20glucose%20and%20insulin%20concentrations%20fell%20acutely%20during%20the%201st%203%20days%20of%20fasting%2C%20and%20alpha%20amino%20nitrogen%20after%2017%20days.%20The%20concentration%20of%20free%20fatty%20acids%2C%20beta-hydroxybutyrate%2C%20and%20acetoacetate%20did%20not%20reach%20a%20plateau%20until%20after%2017%20days.%20Estimated%20glucose%20production%20at%205-6%20wk%20of%20starvation%20is%20reduced%20to%20approximately%2086%20g%5C%2F24%20hr.%20Of%20this%20amount%20the%20liver%20contributes%20about%20one-half%20and%20the%20kidney%20the%20remainder.%20Approximately%20all%20of%20the%20lactate%2C%20pyruvate%2C%20glycerol%2C%20and%20amino%20acid%20carbons%20which%20are%20removed%20by%20liver%20and%20kidney%20are%20converted%20into%20glucose%2C%20as%20evidenced%20by%20substrate%20balances%20across%20these%20organs.%22%2C%22date%22%3A%22Mar%201969%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1172%5C%2FJCI106016%22%2C%22ISSN%22%3A%220021-9738%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22KMY5B6AS%22%2C%22U6SB2T82%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A01%3A55Z%22%7D%7D%2C%7B%22key%22%3A%22ES9NPU2S%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Carlson%20and%20Hoelzel%22%2C%22parsedDate%22%3A%221946%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ECarlson%20AJ%2C%20Hoelzel%20F%20%281946%29%20Apparent%20prolongation%20of%20the%20life%20span%20of%20rats%20by%20intermittent%20fasting.%20J%20Nutr%2031%3A363%26%23x2013%3B375.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1093%5C%2Fjn%5C%2F31.3.363%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1093%5C%2Fjn%5C%2F31.3.363%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Apparent%20prolongation%20of%20the%20life%20span%20of%20rats%20by%20intermittent%20fasting%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%20J.%22%2C%22lastName%22%3A%22Carlson%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22F.%22%2C%22lastName%22%3A%22Hoelzel%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%2203%201946%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1093%5C%2Fjn%5C%2F31.3.363%22%2C%22ISSN%22%3A%220022-3166%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22HXG2U5JG%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A01%3A43Z%22%7D%7D%2C%7B%22key%22%3A%22CE2PQYYN%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Carmona-Gutierrez%20et%20al.%22%2C%22parsedDate%22%3A%222019%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ECarmona-Gutierrez%20D%2C%20Zimmermann%20A%2C%20Kainz%20K%2C%20et%20al%20%282019%29%20The%20flavonoid%204%2C4%26%23x2019%3B-dimethoxychalcone%20promotes%20autophagy-dependent%20longevity%20across%20species.%20Nat%20Commun%2010%3A651.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1038%5C%2Fs41467-019-08555-w%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1038%5C%2Fs41467-019-08555-w%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22The%20flavonoid%204%2C4%27-dimethoxychalcone%20promotes%20autophagy-dependent%20longevity%20across%20species%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Didac%22%2C%22lastName%22%3A%22Carmona-Gutierrez%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Andreas%22%2C%22lastName%22%3A%22Zimmermann%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katharina%22%2C%22lastName%22%3A%22Kainz%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Federico%22%2C%22lastName%22%3A%22Pietrocola%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guo%22%2C%22lastName%22%3A%22Chen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Silvia%22%2C%22lastName%22%3A%22Maglioni%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Alfonso%22%2C%22lastName%22%3A%22Schiavi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jihoon%22%2C%22lastName%22%3A%22Nah%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sara%22%2C%22lastName%22%3A%22Mertel%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christine%20B.%22%2C%22lastName%22%3A%22Beuschel%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Francesca%22%2C%22lastName%22%3A%22Castoldi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Valentina%22%2C%22lastName%22%3A%22Sica%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gert%22%2C%22lastName%22%3A%22Trausinger%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Reingard%22%2C%22lastName%22%3A%22Raml%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Cornelia%22%2C%22lastName%22%3A%22Sommer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sabrina%22%2C%22lastName%22%3A%22Schroeder%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastian%20J.%22%2C%22lastName%22%3A%22Hofer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Maria%20A.%22%2C%22lastName%22%3A%22Bauer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tobias%22%2C%22lastName%22%3A%22Pendl%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jelena%22%2C%22lastName%22%3A%22Tadic%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christopher%22%2C%22lastName%22%3A%22Dammbrueck%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Zehan%22%2C%22lastName%22%3A%22Hu%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christoph%22%2C%22lastName%22%3A%22Ruckenstuhl%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tobias%22%2C%22lastName%22%3A%22Eisenberg%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sylvere%22%2C%22lastName%22%3A%22Durand%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22No%5Cu00e9lie%22%2C%22lastName%22%3A%22Bossut%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fanny%22%2C%22lastName%22%3A%22Aprahamian%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mahmoud%22%2C%22lastName%22%3A%22Abdellatif%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Simon%22%2C%22lastName%22%3A%22Sedej%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22David%20P.%22%2C%22lastName%22%3A%22Enot%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Heimo%22%2C%22lastName%22%3A%22Wolinski%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22J%5Cu00f6rn%22%2C%22lastName%22%3A%22Dengjel%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Oliver%22%2C%22lastName%22%3A%22Kepp%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christoph%22%2C%22lastName%22%3A%22Magnes%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Frank%22%2C%22lastName%22%3A%22Sinner%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Thomas%20R.%22%2C%22lastName%22%3A%22Pieber%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Junichi%22%2C%22lastName%22%3A%22Sadoshima%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Natascia%22%2C%22lastName%22%3A%22Ventura%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Stephan%20J.%22%2C%22lastName%22%3A%22Sigrist%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Kroemer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Frank%22%2C%22lastName%22%3A%22Madeo%22%7D%5D%2C%22abstractNote%22%3A%22Ageing%20constitutes%20the%20most%20important%20risk%20factor%20for%20all%20major%20chronic%20ailments%2C%20including%20malignant%2C%20cardiovascular%20and%20neurodegenerative%20diseases.%20However%2C%20behavioural%20and%20pharmacological%20interventions%20with%20feasible%20potential%20to%20promote%20health%20upon%20ageing%20remain%20rare.%20Here%20we%20report%20the%20identification%20of%20the%20flavonoid%204%2C4%27-dimethoxychalcone%20%28DMC%29%20as%20a%20natural%20compound%20with%20anti-ageing%20properties.%20External%20DMC%20administration%20extends%20the%20lifespan%20of%20yeast%2C%20worms%20and%20flies%2C%20decelerates%20senescence%20of%20human%20cell%20cultures%2C%20and%20protects%20mice%20from%20prolonged%20myocardial%20ischaemia.%20Concomitantly%2C%20DMC%20induces%20autophagy%2C%20which%20is%20essential%20for%20its%20cytoprotective%20effects%20from%20yeast%20to%20mice.%20This%20pro-autophagic%20response%20induces%20a%20conserved%20systemic%20change%20in%20metabolism%2C%20operates%20independently%20of%20TORC1%20signalling%20and%20depends%20on%20specific%20GATA%20transcription%20factors.%20Notably%2C%20we%20identify%20DMC%20in%20the%20plant%20Angelica%20keiskei%20koidzumi%2C%20to%20which%20longevity-%20and%20health-promoting%20effects%20are%20ascribed%20in%20Asian%20traditional%20medicine.%20In%20summary%2C%20we%20have%20identified%20and%20mechanistically%20characterised%20the%20conserved%20longevity-promoting%20effects%20of%20a%20natural%20anti-ageing%20drug.%22%2C%22date%22%3A%2202%2019%2C%202019%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1038%5C%2Fs41467-019-08555-w%22%2C%22ISSN%22%3A%222041-1723%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22FM3XX4QZ%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A00%3A34Z%22%7D%7D%2C%7B%22key%22%3A%22GJB9K3L2%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Caccialanza%20et%20al.%22%2C%22parsedDate%22%3A%222019%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ECaccialanza%20R%2C%20Aprile%20G%2C%20Cereda%20E%2C%20Pedrazzoli%20P%20%282019%29%20Fasting%20in%20oncology%3A%20a%20word%20of%20caution.%20Nat%20Rev%20Cancer%2019%3A177.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1038%5C%2Fs41568-018-0098-0%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1038%5C%2Fs41568-018-0098-0%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Fasting%20in%20oncology%3A%20a%20word%20of%20caution%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Riccardo%22%2C%22lastName%22%3A%22Caccialanza%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Giuseppe%22%2C%22lastName%22%3A%22Aprile%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Emanuele%22%2C%22lastName%22%3A%22Cereda%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Paolo%22%2C%22lastName%22%3A%22Pedrazzoli%22%7D%5D%2C%22abstractNote%22%3A%22%22%2C%22date%22%3A%2203%202019%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1038%5C%2Fs41568-018-0098-0%22%2C%22ISSN%22%3A%221474-1768%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22G537RKA2%22%5D%2C%22dateModified%22%3A%222019-06-18T06%3A00%3A14Z%22%7D%7D%2C%7B%22key%22%3A%22XTBGIK8S%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Pietrocola%20et%20al.%22%2C%22parsedDate%22%3A%222017-03-04%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EPietrocola%20F%2C%20Demont%20Y%2C%20Castoldi%20F%2C%20et%20al%20%282017%29%20Metabolic%20effects%20of%20fasting%20on%20human%20and%20mouse%20blood%20in%20vivo.%20Autophagy%2013%3A567%26%23x2013%3B578.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1080%5C%2F15548627.2016.1271513%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1080%5C%2F15548627.2016.1271513%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Metabolic%20effects%20of%20fasting%20on%20human%20and%20mouse%20blood%20in%20vivo%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Federico%22%2C%22lastName%22%3A%22Pietrocola%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yohann%22%2C%22lastName%22%3A%22Demont%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Francesca%22%2C%22lastName%22%3A%22Castoldi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22David%22%2C%22lastName%22%3A%22Enot%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sylv%5Cu00e8re%22%2C%22lastName%22%3A%22Durand%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Michaela%22%2C%22lastName%22%3A%22Semeraro%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Elisa%20Elena%22%2C%22lastName%22%3A%22Baracco%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jonathan%22%2C%22lastName%22%3A%22Pol%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jose%20Manuel%22%2C%22lastName%22%3A%22Bravo-San%20Pedro%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Chlo%5Cu00e9%22%2C%22lastName%22%3A%22Bordenave%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sarah%22%2C%22lastName%22%3A%22Levesque%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Juliette%22%2C%22lastName%22%3A%22Humeau%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Alexis%22%2C%22lastName%22%3A%22Chery%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Didier%22%2C%22lastName%22%3A%22M%5Cu00e9tivier%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Frank%22%2C%22lastName%22%3A%22Madeo%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%20Chiara%22%2C%22lastName%22%3A%22Maiuri%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Kroemer%22%7D%5D%2C%22abstractNote%22%3A%22Starvation%20is%20a%20strong%20physiological%20stimulus%20of%20macroautophagy%5C%2Fautophagy.%20In%20this%20study%2C%20we%20addressed%20the%20question%20as%20to%20whether%20it%20would%20be%20possible%20to%20measure%20autophagy%20in%20blood%20cells%20after%20nutrient%20deprivation.%20Fasting%20of%20mice%20for%2048%5Cu00a0h%20%28which%20causes%20%5Cu223c20%25%20weight%20loss%29%20or%20starvation%20of%20human%20volunteers%20for%20up%20to%204%5Cu00a0d%20%28which%20causes%20%3C2%25%20weight%20loss%29%20provokes%20major%20changes%20in%20the%20plasma%20metabolome%2C%20yet%20induces%20only%20relatively%20minor%20alterations%20in%20the%20intracellular%20metabolome%20of%20circulating%20leukocytes.%20White%20blood%20cells%20from%20mice%20and%20human%20volunteers%20responded%20to%20fasting%20with%20a%20marked%20reduction%20in%20protein%20lysine%20acetylation%2C%20affecting%20both%20nuclear%20and%20cytoplasmic%20compartments.%20In%20circulating%20leukocytes%20from%20mice%20that%20underwent%2048-h%20fasting%2C%20an%20increase%20in%20LC3B%20lipidation%20%28as%20assessed%20by%20immunoblotting%20and%20immunofluorescence%29%20only%20became%20detectable%20if%20the%20protease%20inhibitor%20leupeptin%20was%20injected%202%5Cu00a0h%20before%20drawing%20blood.%20Consistently%2C%20measurement%20of%20an%20enhanced%20autophagic%20flux%20was%20only%20possible%20if%20white%20blood%20cells%20from%20starved%20human%20volunteers%20were%20cultured%20in%20the%20presence%20or%20absence%20of%20leupeptin.%20Whereas%20all%20murine%20leukocyte%20subpopulations%20significantly%20increased%20the%20number%20of%20LC3B%2B%20puncta%20per%20cell%20in%20response%20to%20nutrient%20deprivation%2C%20only%20neutrophils%20from%20starved%20volunteers%20showed%20signs%20of%20activated%20autophagy%20%28as%20determined%20by%20a%20combination%20of%20multi-color%20immunofluorescence%2C%20cytofluorometry%20and%20image%20analysis%29.%20Altogether%2C%20these%20results%20suggest%20that%20white%20blood%20cells%20are%20suitable%20for%20monitoring%20autophagic%20flux.%20In%20addition%2C%20we%20propose%20that%20the%20evaluation%20of%20protein%20acetylation%20in%20circulating%20leukocytes%20can%20be%20adopted%20as%20a%20biochemical%20marker%20of%20organismal%20energetic%20status.%22%2C%22date%22%3A%22Mar%2004%2C%202017%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1080%5C%2F15548627.2016.1271513%22%2C%22ISSN%22%3A%221554-8635%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22FM3XX4QZ%22%2C%2247PY442C%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A58%3A51Z%22%7D%7D%2C%7B%22key%22%3A%224NB77YNV%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Schultz%20and%20Sinclair%22%2C%22parsedDate%22%3A%222016-01-01%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ESchultz%20MB%2C%20Sinclair%20DA%20%282016%29%20When%20stem%20cells%20grow%20old%3A%20phenotypes%20and%20mechanisms%20of%20stem%20cell%20aging.%20Development%20143%3A3%26%23x2013%3B14.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1242%5C%2Fdev.130633%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1242%5C%2Fdev.130633%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22When%20stem%20cells%20grow%20old%3A%20phenotypes%20and%20mechanisms%20of%20stem%20cell%20aging%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Michael%20B.%22%2C%22lastName%22%3A%22Schultz%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22David%20A.%22%2C%22lastName%22%3A%22Sinclair%22%7D%5D%2C%22abstractNote%22%3A%22All%20multicellular%20organisms%20undergo%20a%20decline%20in%20tissue%20and%20organ%20function%20as%20they%20age.%20An%20attractive%20theory%20is%20that%20a%20loss%20in%20stem%20cell%20number%20and%5C%2For%20activity%20over%20time%20causes%20this%20decline.%20In%20accordance%20with%20this%20theory%2C%20aging%20phenotypes%20have%20been%20described%20for%20stem%20cells%20of%20multiple%20tissues%2C%20including%20those%20of%20the%20hematopoietic%20system%2C%20intestine%2C%20muscle%2C%20brain%2C%20skin%20and%20germline.%20Here%2C%20we%20discuss%20recent%20advances%20in%20our%20understanding%20of%20why%20adult%20stem%20cells%20age%20and%20how%20this%20aging%20impacts%20diseases%20and%20lifespan.%20With%20this%20increased%20understanding%2C%20it%20is%20feasible%20to%20design%20and%20test%20interventions%20that%20delay%20stem%20cell%20aging%20and%20improve%20both%20health%20and%20lifespan.%22%2C%22date%22%3A%22Jan%2001%2C%202016%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1242%5C%2Fdev.130633%22%2C%22ISSN%22%3A%221477-9129%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22FXC3FTC4%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A58%3A42Z%22%7D%7D%2C%7B%22key%22%3A%22ZZT2RDW2%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Dorff%20et%20al.%22%2C%22parsedDate%22%3A%222016%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EDorff%20TB%2C%20Groshen%20S%2C%20Garcia%20A%2C%20et%20al%20%282016%29%20Safety%20and%20feasibility%20of%20fasting%20in%20combination%20with%20platinum-based%20chemotherapy.%20BMC%20Cancer%2016%3A360.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1186%5C%2Fs12885-016-2370-6%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1186%5C%2Fs12885-016-2370-6%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Safety%20and%20feasibility%20of%20fasting%20in%20combination%20with%20platinum-based%20chemotherapy%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tanya%20B.%22%2C%22lastName%22%3A%22Dorff%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Susan%22%2C%22lastName%22%3A%22Groshen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Agustin%22%2C%22lastName%22%3A%22Garcia%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Manali%22%2C%22lastName%22%3A%22Shah%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Denice%22%2C%22lastName%22%3A%22Tsao-Wei%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Huyen%22%2C%22lastName%22%3A%22Pham%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Chia-Wei%22%2C%22lastName%22%3A%22Cheng%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sebastian%22%2C%22lastName%22%3A%22Brandhorst%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pinchas%22%2C%22lastName%22%3A%22Cohen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Min%22%2C%22lastName%22%3A%22Wei%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Valter%22%2C%22lastName%22%3A%22Longo%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22David%20I.%22%2C%22lastName%22%3A%22Quinn%22%7D%5D%2C%22abstractNote%22%3A%22BACKGROUND%3A%20Short-term%20starvation%20prior%20to%20chemotherapy%20administration%20protects%20mice%20against%20toxicity.%20We%20undertook%20dose-escalation%20of%20fasting%20prior%20to%20platinum-based%20chemotherapy%20to%20determine%20safety%20and%20feasibility%20in%20cancer%20patients.%5CnMETHODS%3A%203%20cohorts%20fasted%20before%20chemotherapy%20for%2024%2C%2048%20and%2072%5Cu00a0h%20%28divided%20as%2048%20pre-chemo%20and%2024%20post-chemo%29%20and%20recorded%20all%20calories%20consumed.%20Feasibility%20was%20defined%20as%5Cu2009%5Cu2265%5Cu20093%5C%2F6%20subjects%20in%20each%20cohort%20consuming%5Cu2009%5Cu2264%5Cu2009200%5Cu00a0kcal%20per%2024%5Cu00a0h%20during%20the%20fast%20period%20without%20excess%20toxicity.%20Oxidative%20stress%20was%20evaluated%20in%20leukocytes%20using%20the%20COMET%20assay.%20Insulin%2C%20glucose%2C%20ketones%2C%20insulin-like%20growth%20factor-1%20%28IGF-1%29%20and%20IGF%20binding%20proteins%20%28IGFBPs%29%20were%20measured%20as%20biomarkers%20of%20the%20fasting%20state.%5CnRESULTS%3A%20The%20median%20age%20of%20our%2020%20subjects%20was%2061%2C%20and%2085%5Cu00a0%25%20were%20women.%20Feasibility%20criteria%20were%20met.%20Fasting-related%20toxicities%20were%20limited%20to%5Cu2009%5Cu2264%5Cu2009grade%202%2C%20most%20commonly%20fatigue%2C%20headache%2C%20and%20dizziness.%20The%20COMET%20assay%20indicated%20reduced%20DNA%20damage%20in%20leukocytes%20from%20subjects%20who%20fasted%20for%20%5Cu226548%5Cu00a0h%20%28p%5Cu2009%3D%5Cu20090.08%29.%20There%20was%20a%20non-significant%20trend%20toward%20less%20grade%203%20or%204%20neutropenia%20in%20the%2048%20and%2072%5Cu00a0h%20cohorts%20compared%20to%2024%5Cu00a0h%20cohort%20%28p%5Cu2009%3D%5Cu20090.17%29.%20IGF-1%20levels%20decreased%20by%2030%2C%2033%20and%208%5Cu00a0%25%20in%20the%2024%2C%2048%20and%2072%5Cu00a0h%20fasting%20cohorts%20respectively%20after%20the%20first%20fasting%20period.%5CnCONCLUSION%3A%20Fasting%20for%2072%5Cu00a0h%20around%20chemotherapy%20administration%20is%20safe%20and%20feasible%20for%20cancer%20patients.%20Biomarkers%20such%20as%20IGF-1%20may%20facilitate%20assessment%20of%20differences%20in%20chemotherapy%20toxicity%20in%20subgroups%20achieving%20the%20physiologic%20fasting%20state.%20An%20onging%20randomized%20trial%20is%20studying%20the%20effect%20of%2072%5Cu00a0h%20of%20fasting.%5CnTRIAL%20REGISTRATION%3A%20NCT00936364%20%2C%20registered%20propectively%20on%20July%209%2C%202009.%22%2C%22date%22%3A%2206%2010%2C%202016%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1186%5C%2Fs12885-016-2370-6%22%2C%22ISSN%22%3A%221471-2407%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%2247PY442C%22%2C%22G537RKA2%22%2C%22ANWER6MZ%22%2C%224ZIJ8T4Y%22%2C%22KBEI2USB%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A58%3A32Z%22%7D%7D%2C%7B%22key%22%3A%223XRK86DQ%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Vendelbo%20et%20al.%22%2C%22parsedDate%22%3A%222014%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EVendelbo%20MH%2C%20M%26%23xF8%3Bller%20AB%2C%20Christensen%20B%2C%20et%20al%20%282014%29%20Fasting%20increases%20human%20skeletal%20muscle%20net%20phenylalanine%20release%20and%20this%20is%20associated%20with%20decreased%20mTOR%20signaling.%20PLoS%20ONE%209%3Ae102031.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1371%5C%2Fjournal.pone.0102031%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1371%5C%2Fjournal.pone.0102031%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Fasting%20increases%20human%20skeletal%20muscle%20net%20phenylalanine%20release%20and%20this%20is%20associated%20with%20decreased%20mTOR%20signaling%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mikkel%20Holm%22%2C%22lastName%22%3A%22Vendelbo%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Andreas%20Buch%22%2C%22lastName%22%3A%22M%5Cu00f8ller%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Britt%22%2C%22lastName%22%3A%22Christensen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Birgitte%22%2C%22lastName%22%3A%22Nellemann%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Berthil%20Frederik%20Forrest%22%2C%22lastName%22%3A%22Clasen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22K.%20Sreekumaran%22%2C%22lastName%22%3A%22Nair%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jens%20Otto%20Lunde%22%2C%22lastName%22%3A%22J%5Cu00f8rgensen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Niels%22%2C%22lastName%22%3A%22Jessen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Niels%22%2C%22lastName%22%3A%22M%5Cu00f8ller%22%7D%5D%2C%22abstractNote%22%3A%22AIM%3A%20Fasting%20is%20characterised%20by%20profound%20changes%20in%20energy%20metabolism%20including%20progressive%20loss%20of%20body%20proteins.%20The%20underlying%20mechanisms%20are%20however%20unknown%20and%20we%20therefore%20determined%20the%20effects%20of%20a%2072-hour-fast%20on%20human%20skeletal%20muscle%20protein%20metabolism%20and%20activation%20of%20mammalian%20target%20of%20rapamycin%20%28mTOR%29%2C%20a%20key%20regulator%20of%20cell%20growth.%5CnMETHODS%3A%20Eight%20healthy%20male%20volunteers%20were%20studied%20twice%3A%20in%20the%20postabsorptive%20state%20and%20following%2072%20hours%20of%20fasting.%20Regional%20muscle%20amino%20acid%20kinetics%20was%20measured%20in%20the%20forearm%20using%20amino%20acid%20tracers.%20Signaling%20to%20protein%20synthesis%20and%20breakdown%20were%20assessed%20in%20skeletal%20muscle%20biopsies%20obtained%20during%20non-insulin%20and%20insulin%20stimulated%20conditions%20on%20both%20examination%20days.%5CnRESULTS%3A%20Fasting%20significantly%20increased%20forearm%20net%20phenylalanine%20release%20and%20tended%20to%20decrease%20phenylalanine%20rate%20of%20disappearance.%20mTOR%20phosphorylation%20was%20decreased%20by%20%5Cu223c50%25%20following%20fasting%2C%20together%20with%20reduced%20downstream%20phosphorylation%20of%204EBP1%2C%20ULK1%20and%20rpS6.%20In%20addition%2C%20the%20insulin%20stimulated%20increase%20in%20mTOR%20and%20rpS6%20phosphorylation%20was%20significantly%20reduced%20after%20fasting%20indicating%20insulin%20resistance%20in%20this%20part%20of%20the%20signaling%20pathway.%20Autophagy%20initiation%20is%20in%20part%20regulated%20by%20mTOR%20through%20ULK1%20and%20fasting%20increased%20expression%20of%20the%20autophagic%20marker%20LC3B-II%20by%20%5Cu223c30%25.%20p62%20is%20degraded%20during%20autophagy%20but%20was%20increased%20by%20%5Cu223c10%25%20during%20fasting%20making%20interpretation%20of%20autophagic%20flux%20problematic.%20MAFbx%20and%20MURF1%20ubiquitin%20ligases%20remained%20unaltered%20after%20fasting%20indicating%20no%20change%20in%20protesomal%20protein%20degradation.%5CnCONCLUSIONS%3A%20Our%20results%20show%20that%20during%20fasting%20increased%20net%20phenylalanine%20release%20in%20skeletal%20muscle%20is%20associated%20to%20reduced%20mTOR%20activation%20and%20concomitant%20decreased%20downstream%20signaling%20to%20cell%20growth.%22%2C%22date%22%3A%222014%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1371%5C%2Fjournal.pone.0102031%22%2C%22ISSN%22%3A%221932-6203%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22FM3XX4QZ%22%2C%22KMY5B6AS%22%2C%2247PY442C%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A57%3A59Z%22%7D%7D%2C%7B%22key%22%3A%22TWZWWXS5%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Ezaki%20et%20al.%22%2C%22parsedDate%22%3A%222011-07%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EEzaki%20J%2C%20Matsumoto%20N%2C%20Takeda-Ezaki%20M%2C%20et%20al%20%282011%29%20Liver%20autophagy%20contributes%20to%20the%20maintenance%20of%20blood%20glucose%20and%20amino%20acid%20levels.%20Autophagy%207%3A727%26%23x2013%3B736.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.4161%5C%2Fauto.7.7.15371%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.4161%5C%2Fauto.7.7.15371%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Liver%20autophagy%20contributes%20to%20the%20maintenance%20of%20blood%20glucose%20and%20amino%20acid%20levels%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Junji%22%2C%22lastName%22%3A%22Ezaki%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Naomi%22%2C%22lastName%22%3A%22Matsumoto%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsue%22%2C%22lastName%22%3A%22Takeda-Ezaki%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Masaaki%22%2C%22lastName%22%3A%22Komatsu%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Katsuyuki%22%2C%22lastName%22%3A%22Takahashi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Yuka%22%2C%22lastName%22%3A%22Hiraoka%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hikari%22%2C%22lastName%22%3A%22Taka%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tsutomu%22%2C%22lastName%22%3A%22Fujimura%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kenji%22%2C%22lastName%22%3A%22Takehana%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mitsutaka%22%2C%22lastName%22%3A%22Yoshida%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Junichi%22%2C%22lastName%22%3A%22Iwata%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Isei%22%2C%22lastName%22%3A%22Tanida%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Norihiko%22%2C%22lastName%22%3A%22Furuya%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Dong-Mei%22%2C%22lastName%22%3A%22Zheng%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Norihiro%22%2C%22lastName%22%3A%22Tada%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Keiji%22%2C%22lastName%22%3A%22Tanaka%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eiki%22%2C%22lastName%22%3A%22Kominami%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Takashi%22%2C%22lastName%22%3A%22Ueno%22%7D%5D%2C%22abstractNote%22%3A%22Both%20anabolism%20and%20catabolism%20of%20the%20amino%20acids%20released%20by%20starvation-induced%20autophagy%20are%20essential%20for%20cell%20survival%2C%20but%20their%20actual%20metabolic%20contributions%20in%20adult%20animals%20are%20poorly%20understood.%20Herein%2C%20we%20report%20that%2C%20in%20mice%2C%20liver%20autophagy%20makes%20a%20significant%20contribution%20to%20the%20maintenance%20of%20blood%20glucose%20by%20converting%20amino%20acids%20to%20glucose%20via%20gluconeogenesis.%20Under%20a%20synchronous%20fasting-initiation%20regimen%2C%20autophagy%20was%20induced%20concomitantly%20with%20a%20fall%20in%20plasma%20insulin%20in%20the%20presence%20of%20stable%20glucagon%20levels%2C%20resulting%20in%20a%20robust%20amino%20acid%20release.%20In%20liver-specific%20autophagy%20%28Atg7%29-deficient%20mice%2C%20no%20amino%20acid%20release%20occurred%20and%20blood%20glucose%20levels%20continued%20to%20decrease%20in%20contrast%20to%20those%20of%20wild-type%20mice.%20Administration%20of%20serine%20%2830%20mg%5C%2Fanimal%29%20exerted%20a%20comparable%20effect%2C%20raising%20the%20blood%20glucose%20levels%20in%20both%20control%20wild-type%20and%20mutant%20mice%20under%20starvation.%20Thus%2C%20the%20absence%20of%20the%20amino%20acids%20that%20were%20released%20by%20autophagic%20proteolysis%20is%20a%20major%20reason%20for%20a%20decrease%20in%20blood%20glucose.%20Autophagic%20amino%20acid%20release%20in%20control%20wild-type%20livers%20was%20significantly%20suppressed%20by%20the%20prior%20administration%20of%20glucose%2C%20which%20elicited%20a%20prompt%20increase%20in%20plasma%20insulin%20levels.%20This%20indicates%20that%20insulin%20plays%20a%20dominant%20role%20over%20glucagon%20in%20controlling%20liver%20autophagy.%20These%20results%20are%20the%20first%20to%20show%20that%20liver-specific%20autophagy%20plays%20a%20role%20in%20blood%20glucose%20regulation.%22%2C%22date%22%3A%22Jul%202011%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.4161%5C%2Fauto.7.7.15371%22%2C%22ISSN%22%3A%221554-8635%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22FM3XX4QZ%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A57%3A33Z%22%7D%7D%2C%7B%22key%22%3A%22XL67M6CI%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Dykstra%20et%20al.%22%2C%22parsedDate%22%3A%222011-12-19%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EDykstra%20B%2C%20Olthof%20S%2C%20Schreuder%20J%2C%20et%20al%20%282011%29%20Clonal%20analysis%20reveals%20multiple%20functional%20defects%20of%20aged%20murine%20hematopoietic%20stem%20cells.%20J%20Exp%20Med%20208%3A2691%26%23x2013%3B2703.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1084%5C%2Fjem.20111490%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1084%5C%2Fjem.20111490%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Clonal%20analysis%20reveals%20multiple%20functional%20defects%20of%20aged%20murine%20hematopoietic%20stem%20cells%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Brad%22%2C%22lastName%22%3A%22Dykstra%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sandra%22%2C%22lastName%22%3A%22Olthof%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jaring%22%2C%22lastName%22%3A%22Schreuder%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Martha%22%2C%22lastName%22%3A%22Ritsema%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gerald%22%2C%22lastName%22%3A%22de%20Haan%22%7D%5D%2C%22abstractNote%22%3A%22Hematopoietic%20stem%20cell%20%28HSC%29%20populations%20change%20with%20aging%2C%20but%20the%20extent%20to%20which%20this%20is%20caused%20by%20qualitative%20versus%20quantitative%20alterations%20in%20HSC%20subtypes%20is%20unclear.%20Using%20clonal%20assays%2C%20in%20this%20study%20we%20show%20that%20the%20aging%20HSC%20compartment%20undergoes%20both%20quantitative%20and%20qualitative%20changes.%20We%20observed%20a%20variable%20increase%20of%20HSC%20pool%20size%20with%20age%2C%20accompanied%20by%20the%20accumulation%20of%20predominantly%20myeloid-biased%20HSCs%20that%20regenerate%20substantially%20fewer%20mature%20progeny%20than%20young%20myeloid-biased%20HSCs%20and%20exhibit%20reduced%20self-renewal%20activity%20as%20measured%20by%20long-term%20secondary%20transplantation.%20Old%20HSCs%20had%20a%20twofold%20reduction%20in%20marrow-homing%20efficiency%20and%20a%20similar%20decrease%20in%20functional%20frequency%20as%20measured%20using%20long-term%20transplantation%20assays.%20Similarly%2C%20old%20HSCs%20had%20a%20twofold%20reduced%20seeding%20efficiency%20and%20a%20significantly%20delayed%20proliferative%20response%20compared%20with%20young%20HSCs%20in%20long-term%20stromal%20cell%20co-cultures%20but%20were%20indistinguishable%20in%20suspension%20cultures.%20We%20show%20that%20these%20functional%20defects%20are%20characteristics%20of%20most%20or%20all%20old%20HSCs%20and%20are%20not%20indicative%20of%20a%20nonfunctional%20subset%20of%20cells%20that%20express%20HSC%20markers.%20Furthermore%2C%20we%20demonstrate%20that%20cells%20with%20functional%20properties%20of%20old%20HSCs%20can%20be%20generated%20directly%20from%20young%20HSCs%20by%20extended%20serial%20transplantation%2C%20which%20is%20consistent%20with%20the%20possibility%20that%20they%20arise%20through%20a%20process%20of%20cellular%20aging.%22%2C%22date%22%3A%22Dec%2019%2C%202011%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1084%5C%2Fjem.20111490%22%2C%22ISSN%22%3A%221540-9538%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22FXC3FTC4%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A57%3A27Z%22%7D%7D%2C%7B%22key%22%3A%22NCTGYSIU%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Safdie%20et%20al.%22%2C%22parsedDate%22%3A%222009-12-31%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ESafdie%20FM%2C%20Dorff%20T%2C%20Quinn%20D%2C%20et%20al%20%282009%29%20Fasting%20and%20cancer%20treatment%20in%20humans%3A%20A%20case%20series%20report.%20Aging%20%28Albany%20NY%29%201%3A988%26%23x2013%3B1007.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.18632%5C%2Faging.100114%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.18632%5C%2Faging.100114%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Fasting%20and%20cancer%20treatment%20in%20humans%3A%20A%20case%20series%20report%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fernando%20M.%22%2C%22lastName%22%3A%22Safdie%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tanya%22%2C%22lastName%22%3A%22Dorff%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22David%22%2C%22lastName%22%3A%22Quinn%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Luigi%22%2C%22lastName%22%3A%22Fontana%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Min%22%2C%22lastName%22%3A%22Wei%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Changhan%22%2C%22lastName%22%3A%22Lee%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Pinchas%22%2C%22lastName%22%3A%22Cohen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Valter%20D.%22%2C%22lastName%22%3A%22Longo%22%7D%5D%2C%22abstractNote%22%3A%22Short-term%20fasting%20%2848%20hours%29%20was%20shown%20to%20be%20effective%20in%20protecting%20normal%20cells%20and%20mice%20but%20not%20cancer%20cells%20against%20high%20dose%20chemotherapy%2C%20termed%20Differential%20Stress%20Resistance%20%28DSR%29%2C%20but%20the%20feasibility%20and%20effect%20of%20fasting%20in%20cancer%20patients%20undergoing%20chemotherapy%20is%20unknown.%20Here%20we%20describe%2010%20cases%20in%20which%20patients%20diagnosed%20with%20a%20variety%20of%20malignancies%20had%20voluntarily%20fasted%20prior%20to%20%2848-140%20hours%29%20and%5C%2For%20following%20%285-56%20hours%29%20chemotherapy.%20None%20of%20these%20patients%2C%20who%20received%20an%20average%20of%204%20cycles%20of%20various%20chemotherapy%20drugs%20in%20combination%20with%20fasting%2C%20reported%20significant%20side%20effects%20caused%20by%20the%20fasting%20itself%20other%20than%20hunger%20and%20lightheadedness.%20Chemotherapy%20associated%20toxicity%20was%20graded%20according%20to%20the%20Common%20Terminology%20Criteria%20for%20Adverse%20Events%20%28CTCAE%29%20of%20the%20National%20Cancer%20Institute%20%28NCI%29.%20The%20six%20patients%20who%20underwent%20chemotherapy%20with%20or%20without%20fasting%20reported%20a%20reduction%20in%20fatigue%2C%20weakness%2C%20and%20gastrointestinal%20side%20effects%20while%20fasting.%20In%20those%20patients%20whose%20cancer%20progression%20could%20be%20assessed%2C%20fasting%20did%20not%20prevent%20the%20chemotherapy-induced%20reduction%20of%20tumor%20volume%20or%20tumor%20markers.%20Although%20the%2010%20cases%20presented%20here%20suggest%20that%20fasting%20in%20combination%20with%20chemotherapy%20is%20feasible%2C%20safe%2C%20and%20has%20the%20potential%20to%20ameliorate%20side%20effects%20caused%20by%20chemotherapies%2C%20they%20are%20not%20meant%20to%20establish%20practice%20guidelines%20for%20patients%20undergoing%20chemotherapy.%20Only%20controlled-randomized%20clinical%20trials%20will%20determine%20the%20effect%20of%20fasting%20on%20clinical%20outcomes%20including%20quality%20of%20life%20and%20therapeutic%20index.%22%2C%22date%22%3A%22Dec%2031%2C%202009%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.18632%5C%2Faging.100114%22%2C%22ISSN%22%3A%221945-4589%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22KMY5B6AS%22%2C%22G537RKA2%22%2C%22KBEI2USB%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A55%3A38Z%22%7D%7D%2C%7B%22key%22%3A%22AFC6LMAY%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Mathew%20et%20al.%22%2C%22parsedDate%22%3A%222009-06-12%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EMathew%20R%2C%20Karp%20CM%2C%20Beaudoin%20B%2C%20et%20al%20%282009%29%20Autophagy%20suppresses%20tumorigenesis%20through%20elimination%20of%20p62.%20Cell%20137%3A1062%26%23x2013%3B1075.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cell.2009.03.048%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cell.2009.03.048%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Autophagy%20suppresses%20tumorigenesis%20through%20elimination%20of%20p62%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Robin%22%2C%22lastName%22%3A%22Mathew%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Cristina%20M.%22%2C%22lastName%22%3A%22Karp%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Brian%22%2C%22lastName%22%3A%22Beaudoin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Nhan%22%2C%22lastName%22%3A%22Vuong%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guanghua%22%2C%22lastName%22%3A%22Chen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Hsin-Yi%22%2C%22lastName%22%3A%22Chen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kevin%22%2C%22lastName%22%3A%22Bray%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Anupama%22%2C%22lastName%22%3A%22Reddy%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gyan%22%2C%22lastName%22%3A%22Bhanot%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Celine%22%2C%22lastName%22%3A%22Gelinas%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Robert%20S.%22%2C%22lastName%22%3A%22Dipaola%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Vassiliki%22%2C%22lastName%22%3A%22Karantza-Wadsworth%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eileen%22%2C%22lastName%22%3A%22White%22%7D%5D%2C%22abstractNote%22%3A%22Allelic%20loss%20of%20the%20essential%20autophagy%20gene%20beclin1%20occurs%20in%20human%20cancers%20and%20renders%20mice%20tumor-prone%20suggesting%20that%20autophagy%20is%20a%20tumor-suppression%20mechanism.%20While%20tumor%20cells%20utilize%20autophagy%20to%20survive%20metabolic%20stress%2C%20autophagy%20also%20mitigates%20the%20resulting%20cellular%20damage%20that%20may%20limit%20tumorigenesis.%20In%20response%20to%20stress%2C%20autophagy-defective%20tumor%20cells%20preferentially%20accumulated%20p62%5C%2FSQSTM1%20%28p62%29%2C%20endoplasmic%20reticulum%20%28ER%29%20chaperones%2C%20damaged%20mitochondria%2C%20reactive%20oxygen%20species%20%28ROS%29%2C%20and%20genome%20damage.%20Moreover%2C%20suppressing%20ROS%20or%20p62%20accumulation%20prevented%20damage%20resulting%20from%20autophagy%20defects%20indicating%20that%20failure%20to%20regulate%20p62%20caused%20oxidative%20stress.%20Importantly%2C%20sustained%20p62%20expression%20resulting%20from%20autophagy%20defects%20was%20sufficient%20to%20alter%20NF-kappaB%20regulation%20and%20gene%20expression%20and%20to%20promote%20tumorigenesis.%20Thus%2C%20defective%20autophagy%20is%20a%20mechanism%20for%20p62%20upregulation%20commonly%20observed%20in%20human%20tumors%20that%20contributes%20directly%20to%20tumorigenesis%20likely%20by%20perturbing%20the%20signal%20transduction%20adaptor%20function%20of%20p62-controlling%20pathways%20critical%20for%20oncogenesis.%22%2C%22date%22%3A%22Jun%2012%2C%202009%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.cell.2009.03.048%22%2C%22ISSN%22%3A%221097-4172%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22FM3XX4QZ%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A55%3A34Z%22%7D%7D%2C%7B%22key%22%3A%22R2BJ3B97%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Eisenberg%20et%20al.%22%2C%22parsedDate%22%3A%222009-11%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EEisenberg%20T%2C%20Knauer%20H%2C%20Schauer%20A%2C%20et%20al%20%282009%29%20Induction%20of%20autophagy%20by%20spermidine%20promotes%20longevity.%20Nat%20Cell%20Biol%2011%3A1305%26%23x2013%3B1314.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1038%5C%2Fncb1975%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1038%5C%2Fncb1975%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Induction%20of%20autophagy%20by%20spermidine%20promotes%20longevity%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Tobias%22%2C%22lastName%22%3A%22Eisenberg%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Heide%22%2C%22lastName%22%3A%22Knauer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Alexandra%22%2C%22lastName%22%3A%22Schauer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sabrina%22%2C%22lastName%22%3A%22B%5Cu00fcttner%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christoph%22%2C%22lastName%22%3A%22Ruckenstuhl%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Didac%22%2C%22lastName%22%3A%22Carmona-Gutierrez%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Julia%22%2C%22lastName%22%3A%22Ring%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sabrina%22%2C%22lastName%22%3A%22Schroeder%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Christoph%22%2C%22lastName%22%3A%22Magnes%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Lucia%22%2C%22lastName%22%3A%22Antonacci%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Heike%22%2C%22lastName%22%3A%22Fussi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Luiza%22%2C%22lastName%22%3A%22Deszcz%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Regina%22%2C%22lastName%22%3A%22Hartl%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Elisabeth%22%2C%22lastName%22%3A%22Schraml%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Alfredo%22%2C%22lastName%22%3A%22Criollo%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Evgenia%22%2C%22lastName%22%3A%22Megalou%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Daniela%22%2C%22lastName%22%3A%22Weiskopf%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Peter%22%2C%22lastName%22%3A%22Laun%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gino%22%2C%22lastName%22%3A%22Heeren%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Michael%22%2C%22lastName%22%3A%22Breitenbach%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Beatrix%22%2C%22lastName%22%3A%22Grubeck-Loebenstein%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eva%22%2C%22lastName%22%3A%22Herker%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Birthe%22%2C%22lastName%22%3A%22Fahrenkrog%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kai-Uwe%22%2C%22lastName%22%3A%22Fr%5Cu00f6hlich%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Frank%22%2C%22lastName%22%3A%22Sinner%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Nektarios%22%2C%22lastName%22%3A%22Tavernarakis%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Nadege%22%2C%22lastName%22%3A%22Minois%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Kroemer%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Frank%22%2C%22lastName%22%3A%22Madeo%22%7D%5D%2C%22abstractNote%22%3A%22Ageing%20results%20from%20complex%20genetically%20and%20epigenetically%20programmed%20processes%20that%20are%20elicited%20in%20part%20by%20noxious%20or%20stressful%20events%20that%20cause%20programmed%20cell%20death.%20Here%2C%20we%20report%20that%20administration%20of%20spermidine%2C%20a%20natural%20polyamine%20whose%20intracellular%20concentration%20declines%20during%20human%20ageing%2C%20markedly%20extended%20the%20lifespan%20of%20yeast%2C%20flies%20and%20worms%2C%20and%20human%20immune%20cells.%20In%20addition%2C%20spermidine%20administration%20potently%20inhibited%20oxidative%20stress%20in%20ageing%20mice.%20In%20ageing%20yeast%2C%20spermidine%20treatment%20triggered%20epigenetic%20deacetylation%20of%20histone%20H3%20through%20inhibition%20of%20histone%20acetyltransferases%20%28HAT%29%2C%20suppressing%20oxidative%20stress%20and%20necrosis.%20Conversely%2C%20depletion%20of%20endogenous%20polyamines%20led%20to%20hyperacetylation%2C%20generation%20of%20reactive%20oxygen%20species%2C%20early%20necrotic%20death%20and%20decreased%20lifespan.%20The%20altered%20acetylation%20status%20of%20the%20chromatin%20led%20to%20significant%20upregulation%20of%20various%20autophagy-related%20transcripts%2C%20triggering%20autophagy%20in%20yeast%2C%20flies%2C%20worms%20and%20human%20cells.%20Finally%2C%20we%20found%20that%20enhanced%20autophagy%20is%20crucial%20for%20polyamine-induced%20suppression%20of%20necrosis%20and%20enhanced%20longevity.%22%2C%22date%22%3A%22Nov%202009%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1038%5C%2Fncb1975%22%2C%22ISSN%22%3A%221476-4679%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22FM3XX4QZ%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A55%3A30Z%22%7D%7D%2C%7B%22key%22%3A%22VMKSQVPV%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Wheless%22%2C%22parsedDate%22%3A%222008-11%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EWheless%20JW%20%282008%29%20History%20of%20the%20ketogenic%20diet.%20Epilepsia%2049%20Suppl%208%3A3%26%23x2013%3B5.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1111%5C%2Fj.1528-1167.2008.01821.x%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1111%5C%2Fj.1528-1167.2008.01821.x%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22History%20of%20the%20ketogenic%20diet%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22James%20W.%22%2C%22lastName%22%3A%22Wheless%22%7D%5D%2C%22abstractNote%22%3A%22Fasting%20and%20other%20dietary%20regimens%20have%20been%20used%20to%20treat%20epilepsy%20since%20at%20least%20500%20BC.%20To%20mimic%20the%20metabolism%20of%20fasting%2C%20the%20ketogenic%20diet%20%28KD%29%20was%20introduced%20by%20modern%20physicians%20as%20a%20treatment%20for%20epilepsy%20in%20the%201920s.%20For%20two%20decades%20this%20therapy%20was%20widely%20used%2C%20but%20with%20the%20modern%20era%20of%20antiepileptic%20drug%20treatment%20its%20use%20declined%20dramatically.%20By%20the%20end%20of%20the%20twentieth%20century%20this%20therapy%20was%20available%20in%20only%20a%20small%20number%20of%20children%27s%20hospitals.%20Over%20the%20past%2015%20years%2C%20there%20has%20been%20an%20explosion%20in%20the%20use%2C%20and%20scientific%20interest%20in%20the%20KD.%20This%20review%20traces%20the%20history%20of%20one%20of%20the%20most%20effective%20treatments%20for%20childhood%20epilepsy.%22%2C%22date%22%3A%22Nov%202008%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1111%5C%2Fj.1528-1167.2008.01821.x%22%2C%22ISSN%22%3A%221528-1167%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22IB4QNEHC%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A55%3A26Z%22%7D%7D%2C%7B%22key%22%3A%228FYI8Q6H%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Levine%20and%20Kroemer%22%2C%22parsedDate%22%3A%222008-01-11%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ELevine%20B%2C%20Kroemer%20G%20%282008%29%20Autophagy%20in%20the%20pathogenesis%20of%20disease.%20Cell%20132%3A27%26%23x2013%3B42.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cell.2007.12.018%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cell.2007.12.018%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Autophagy%20in%20the%20pathogenesis%20of%20disease%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Beth%22%2C%22lastName%22%3A%22Levine%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Guido%22%2C%22lastName%22%3A%22Kroemer%22%7D%5D%2C%22abstractNote%22%3A%22Autophagy%20is%20a%20lysosomal%20degradation%20pathway%20that%20is%20essential%20for%20survival%2C%20differentiation%2C%20development%2C%20and%20homeostasis.%20Autophagy%20principally%20serves%20an%20adaptive%20role%20to%20protect%20organisms%20against%20diverse%20pathologies%2C%20including%20infections%2C%20cancer%2C%20neurodegeneration%2C%20aging%2C%20and%20heart%20disease.%20However%2C%20in%20certain%20experimental%20disease%20settings%2C%20the%20self-cannibalistic%20or%2C%20paradoxically%2C%20even%20the%20prosurvival%20functions%20of%20autophagy%20may%20be%20deleterious.%20This%20Review%20summarizes%20recent%20advances%20in%20understanding%20the%20physiological%20functions%20of%20autophagy%20and%20its%20possible%20roles%20in%20the%20causation%20and%20prevention%20of%20human%20diseases.%22%2C%22date%22%3A%22Jan%2011%2C%202008%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.cell.2007.12.018%22%2C%22ISSN%22%3A%220092-8674%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22FM3XX4QZ%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A55%3A14Z%22%7D%7D%2C%7B%22key%22%3A%22MF4ZZAX3%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Lee%20et%20al.%22%2C%22parsedDate%22%3A%222006-12%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ELee%20GD%2C%20Wilson%20MA%2C%20Zhu%20M%2C%20et%20al%20%282006%29%20Dietary%20deprivation%20extends%20lifespan%20in%20Caenorhabditis%20elegans.%20Aging%20Cell%205%3A515%26%23x2013%3B524.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1111%5C%2Fj.1474-9726.2006.00241.x%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1111%5C%2Fj.1474-9726.2006.00241.x%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Dietary%20deprivation%20extends%20lifespan%20in%20Caenorhabditis%20elegans%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Garrick%20D.%22%2C%22lastName%22%3A%22Lee%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mark%20A.%22%2C%22lastName%22%3A%22Wilson%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Min%22%2C%22lastName%22%3A%22Zhu%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Catherine%20A.%22%2C%22lastName%22%3A%22Wolkow%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Rafael%22%2C%22lastName%22%3A%22de%20Cabo%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Donald%20K.%22%2C%22lastName%22%3A%22Ingram%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Sige%22%2C%22lastName%22%3A%22Zou%22%7D%5D%2C%22abstractNote%22%3A%22Dietary%20restriction%20%28DR%29%20is%20well%20known%20as%20a%20nongenetic%20intervention%20that%20robustly%20extends%20lifespan%20in%20a%20variety%20of%20species%3B%20however%2C%20its%20underlying%20mechanisms%20remain%20unclear.%20We%20have%20found%20in%20Caenorhabditis%20elegans%20that%20dietary%20deprivation%20%28DD%29%20during%20adulthood%2C%20defined%20as%20removal%20of%20their%20food%20source%20Escherichia%20coli%20after%20the%20completion%20of%20larval%20development%2C%20increased%20lifespan%20and%20enhanced%20thermotolerance%20and%20resistance%20to%20oxidative%20stress.%20DD-induced%20longevity%20was%20independent%20of%20one%20C.%20elegans%20SIRTUIN%2C%20sir-2.1%2C%20which%20is%20required%20for%20the%20effects%20of%20DR%2C%20and%20was%20independent%20of%20the%20daf-2%5C%2Finsulin-like%20signaling%20pathway%20that%20independently%20regulates%20longevity%20and%20larval%20diapause%20in%20C.%20elegans.%20DD%20did%20not%20significantly%20alter%20lifespan%20of%20fem-1%28hc17%29%3B%20eat-2%28ad465%29%20worms%2C%20a%20genetic%20model%20of%20DR.%20These%20findings%20suggest%20that%20DD%20and%20DR%20share%20some%20downstream%20effectors.%20In%20addition%2C%20DD%20was%20detrimental%20for%20longevity%20when%20imposed%20on%20reproductively%20active%20young%20adults%2C%20suggesting%20that%20DD%20may%20only%20be%20beneficial%20in%20the%20absence%20of%20competing%20metabolic%20demands%2C%20such%20as%20fertility.%20Adult-onset%20DD%20offers%20a%20new%20paradigm%20for%20investigating%20dietary%20regulation%20of%20longevity%20in%20C.%20elegans.%20This%20study%20presents%20the%20first%20evidence%20that%20long-term%20DD%2C%20instead%20of%20being%20detrimental%2C%20can%20extend%20lifespan%20of%20a%20multicellular%20adult%20organism.%22%2C%22date%22%3A%22Dec%202006%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1111%5C%2Fj.1474-9726.2006.00241.x%22%2C%22ISSN%22%3A%221474-9718%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22A9H6NEVG%22%2C%22G7863IM7%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A55%3A05Z%22%7D%7D%2C%7B%22key%22%3A%22TYQH7SPE%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Ureta-Vidal%20et%20al.%22%2C%22parsedDate%22%3A%222003-04%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EUreta-Vidal%20A%2C%20Ettwiller%20L%2C%20Birney%20E%20%282003%29%20Comparative%20genomics%3A%20genome-wide%20analysis%20in%20metazoan%20eukaryotes.%20Nat%20Rev%20Genet%204%3A251%26%23x2013%3B262.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1038%5C%2Fnrg1043%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1038%5C%2Fnrg1043%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Comparative%20genomics%3A%20genome-wide%20analysis%20in%20metazoan%20eukaryotes%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Abel%22%2C%22lastName%22%3A%22Ureta-Vidal%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Laurence%22%2C%22lastName%22%3A%22Ettwiller%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ewan%22%2C%22lastName%22%3A%22Birney%22%7D%5D%2C%22abstractNote%22%3A%22The%20increasing%20number%20of%20complete%20and%20nearly%20complete%20metazoan%20genome%20sequences%20provides%20a%20significant%20amount%20of%20material%20for%20large-scale%20comparative%20genomic%20analysis.%20Finding%20new%20effective%20methods%20to%20analyse%20such%20enormous%20datasets%20has%20been%20the%20object%20of%20intense%20research.%20Three%20main%20areas%20in%20comparative%20genomics%20have%20recently%20shown%20important%20developments%3A%20whole-genome%20alignment%2C%20gene%20prediction%20and%20regulatory-region%20prediction.%20Each%20of%20these%20areas%20improves%20the%20methods%20of%20deciphering%20long%20genomic%20sequences%20and%20uncovering%20what%20lies%20hidden%20in%20them.%22%2C%22date%22%3A%22Apr%202003%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1038%5C%2Fnrg1043%22%2C%22ISSN%22%3A%221471-0056%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%227HEDS64Q%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A54%3A54Z%22%7D%7D%2C%7B%22key%22%3A%22QWP452KJ%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Drenick%20et%20al.%22%2C%22parsedDate%22%3A%221972-10%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EDrenick%20EJ%2C%20Alvarez%20LC%2C%20Tamasi%20GC%2C%20Brickman%20AS%20%281972%29%20Resistance%20to%20symptomatic%20insulin%20reactions%20after%20fasting.%20J%20Clin%20Invest%2051%3A2757%26%23x2013%3B2762.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1172%5C%2FJCI107095%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1172%5C%2FJCI107095%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Resistance%20to%20symptomatic%20insulin%20reactions%20after%20fasting%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22E.%20J.%22%2C%22lastName%22%3A%22Drenick%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22L.%20C.%22%2C%22lastName%22%3A%22Alvarez%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22G.%20C.%22%2C%22lastName%22%3A%22Tamasi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%20S.%22%2C%22lastName%22%3A%22Brickman%22%7D%5D%2C%22abstractNote%22%3A%22This%20study%20was%20carried%20out%20to%20determine%20if%2C%20in%20fasting%2C%20an%20adaptation%20to%20utilization%20of%20ketones%20could%20prevent%20cerebral%20dysfunction%20during%20periods%20of%20acute%2C%20insulin-induced%20glucopenia.%20In%20the%20course%20of%20standard%20insulin%20tolerance%20tests%20%280.1-0.2%20U%5C%2Fkg%29%2C%20nine%20obese%20subjects%20manifested%20frank%20hypoglycemic%20reactions%20resulting%20in%20an%20increase%20in%20urinary%20catecholamine%20excretion%20from%2061%20to%20113%20mug%5C%2F24%20hr%20%28P%20%3C%200.01%29.%20After%20fasting%202%20months%2C%20administration%20of%20weight-adjusted%20doses%20of%20insulin%20produced%20identical%20maximum%20insulin%20concentrations%20and%20disappearance%20curves.%20However%2C%20no%20insulin%20reactions%20nor%20significant%20rises%20in%20catecholamine%20excretion%20occurred%20despite%20equal%20extent%20and%20rate%20of%20glucose%20fall.%20Glucose%20concentrations%20as%20low%20as%200.5%20mmoles%5C%2Fliter%20%289%20mg%5C%2F100%20ml%29%20failed%20to%20precipitate%20hypoglycemic%20reactions.%20During%20the%20postfast%20insulin%20tolerance%20tests%2C%20mean%20plasma%202-hydroxybutyrate%20%28beta-OHB%29%20decreased%20from%208.02%20to%206.69%20mmoles%5C%2Fliter%20%28P%20%3C%200.01%29.%20In%20another%20five%20fasting%20subjects%20tested%2C%20the%20A-V%20difference%20for%20beta-OHB%20across%20brain%20increased%20progressively%20from%200.21%20to%200.70%20mmoles%5C%2Fliter%20whereas%20across%20the%20forearm%20no%20consistent%20uptake%20could%20be%20demonstrated.%20Simultaneously%2C%20the%20A-V%20difference%20across%20the%20brain%20for%20glucose%20decreased%20from%200.24%20to%200.07%20mmoles%5C%2Fliter%20of%20plasma.%20In%20addition%20to%20insulin-induced%20suppression%20of%20hepatic%20ketogenesis%2C%20the%20augmented%20cerebral%20ketone%20uptake%20during%20insulin%20hypoglycemia%20contributes%20to%20the%20net%20fall%20in%20plasma%20beta-OHB.%20Ketoacids%2C%20extracted%20by%20the%20fast-adapted%20brain%2C%20supplant%20glucose%20as%20a%20metabolic%20substrate%20preventing%20overt%20hypoglycemic%20reactions%20during%20acute%20glucopenia.%22%2C%22date%22%3A%22Oct%201972%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1172%5C%2FJCI107095%22%2C%22ISSN%22%3A%220021-9738%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22KMY5B6AS%22%5D%2C%22dateModified%22%3A%222019-06-18T05%3A49%3A01Z%22%7D%7D%2C%7B%22key%22%3A%22ZA4XJNFE%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Owen%20et%20al.%22%2C%22parsedDate%22%3A%221967-10%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EOwen%20OE%2C%20Morgan%20AP%2C%20Kemp%20HG%2C%20et%20al%20%281967%29%20Brain%20metabolism%20during%20fasting.%20J%20Clin%20Invest%2046%3A1589%26%23x2013%3B1595.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1172%5C%2FJCI105650%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1172%5C%2FJCI105650%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Brain%20metabolism%20during%20fasting%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22O.%20E.%22%2C%22lastName%22%3A%22Owen%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22A.%20P.%22%2C%22lastName%22%3A%22Morgan%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22H.%20G.%22%2C%22lastName%22%3A%22Kemp%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22J.%20M.%22%2C%22lastName%22%3A%22Sullivan%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22M.%20G.%22%2C%22lastName%22%3A%22Herrera%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22G.%20F.%22%2C%22lastName%22%3A%22Cahill%22%7D%5D%2C%22abstractNote%22%3A%22Catheterization%20of%20cerebral%20vessels%20in%20three%20obese%20patients%20undergoing%205-6%20wk%20of%20starvation%20demonstrated%20that%20beta-hydroxybutyrate%20and%20acetoacetate%20replaced%20glucose%20as%20the%20predominant%20fuel%20for%20brain%20metabolism.%20A%20strikingly%20low%20respiratory%20quotient%20was%20also%20observed%2C%20suggesting%20a%20carboxylation%20mechanism%20as%20a%20means%20of%20disposing%20of%20some%20of%20the%20carbon%20of%20the%20consumed%20substrates.%22%2C%22date%22%3A%22Oct%201967%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1172%5C%2FJCI105650%22%2C%22ISSN%22%3A%220021-9738%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22KMY5B6AS%22%5D%2C%22dateModified%22%3A%222019-06-06T04%3A02%3A21Z%22%7D%7D%2C%7B%22key%22%3A%22S6L3EE64%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22LeCheminant%20et%20al.%22%2C%22parsedDate%22%3A%222013-12-14%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ELeCheminant%20JD%2C%20Christenson%20E%2C%20Bailey%20BW%2C%20Tucker%20LA%20%282013%29%20Restricting%20night-time%20eating%20reduces%20daily%20energy%20intake%20in%20healthy%20young%20men%3A%20a%20short-term%20cross-over%20study.%20Br%20J%20Nutr%20110%3A2108%26%23x2013%3B2113.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1017%5C%2FS0007114513001359%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1017%5C%2FS0007114513001359%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Restricting%20night-time%20eating%20reduces%20daily%20energy%20intake%20in%20healthy%20young%20men%3A%20a%20short-term%20cross-over%20study%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22James%20D.%22%2C%22lastName%22%3A%22LeCheminant%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ed%22%2C%22lastName%22%3A%22Christenson%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Bruce%20W.%22%2C%22lastName%22%3A%22Bailey%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Larry%20A.%22%2C%22lastName%22%3A%22Tucker%22%7D%5D%2C%22abstractNote%22%3A%22Few%20experimental%20data%20are%20available%20to%20support%20the%20notion%20that%20reducing%20night-time%20eating%20changes%20total%20daily%20energy%20intake%20%28EI%29%20or%20body%20weight%20in%20healthy%20adults.%20The%20present%20study%20primarily%20examined%20the%20short-term%20effect%20of%20night%20eating%20restriction%20%28NER%29%20on%20daily%20EI%20in%20healthy%20young%20men.%20It%20secondarily%20examined%20body%20weight%20and%20moods%20associated%20with%20NER.%20Using%20a%20cross-over%20design%2C%20twenty-nine%20men%20%2820%5Cu00b79%20%28sd%202%5Cu00b75%29%20years%3B%2024%5Cu00b74%20%28sd%202%5Cu00b75%29%20kg%5C%2Fm%5Cu00b2%29%20initiated%20a%202-week%20NER%20intervention%20%28elimination%20of%20EI%20from%2019.00%20to%2006.00%20hours%29%20and%20a%202-week%20control%20condition%2C%20counterbalanced%20and%20separated%20by%20a%201-week%20washout%20period.%20EI%20and%20macronutrient%20intake%20were%20assessed%20using%20computerised%2C%20multiple-pass%2024%20h%20food%20recalls%2C%20body%20weight%20via%20a%20digital%20scale%20and%20mood%20using%20the%20Profile%20of%20Mood%20States%20survey.%20Of%20the%20twenty-nine%20participants%2C%20twenty-seven%20%2893%20%25%29%20completed%20all%20aspects%20of%20the%20study.%20During%20the%20NER%20condition%2C%20the%20participants%20consumed%20less%20total%20energy%20per%20d%20than%20during%20the%20control%20condition%20%2810%20125%20v.%2011%20146%20kJ%5C%2Fd%3B%20F%3D%206%5Cu00b741%3B%20P%3D%200%5Cu00b7018%29.%20During%20the%20NER%20condition%2C%20no%20energy%20was%20reported%20consumed%20between%2019.00%20and%2006.00%20hours%3B%20however%2C%20during%20the%20control%20condition%2C%20the%20energy%20intake%20of%20participants%20was%202920%20%28sd%201347%29%20kJ%5C%2Fd%20between%2019.00%20and%2006.00%20hours.%20There%20was%20a%20significant%20difference%20in%20weight%20change%20between%20the%20NER%20%28-0%5Cu00b74%20%28sd%201%5Cu00b71%29%20kg%29%20and%20control%20%28%2B0%5Cu00b76%20%28sd%200%5Cu00b79%29%20kg%29%20conditions%20%28F%3D%2022%5Cu00b768%3B%20P%3C%200%5Cu00b7001%29.%20Differences%20in%20total%20mood%20score%20or%20mood%20subscales%20between%20the%20NER%20and%20control%20conditions%20were%20not%20apparent%20%28P%3E0%5Cu00b705%29.%20These%20findings%20provide%20support%20for%20NER%20decreasing%20short-term%20EI%20in%20healthy%20young%20men.%22%2C%22date%22%3A%22Dec%2014%2C%202013%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1017%5C%2FS0007114513001359%22%2C%22ISSN%22%3A%221475-2662%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22UIM8YXRX%22%2C%226BPRM5LB%22%2C%22JQMRJMTS%22%2C%227WXBJPRG%22%5D%2C%22dateModified%22%3A%222019-06-06T03%3A50%3A24Z%22%7D%7D%2C%7B%22key%22%3A%224NRKPFQR%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Sutton%20et%20al.%22%2C%22parsedDate%22%3A%222018%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ESutton%20EF%2C%20Beyl%20R%2C%20Early%20KS%2C%20et%20al%20%282018%29%20Early%20Time-Restricted%20Feeding%20Improves%20Insulin%20Sensitivity%2C%20Blood%20Pressure%2C%20and%20Oxidative%20Stress%20Even%20without%20Weight%20Loss%20in%20Men%20with%20Prediabetes.%20Cell%20Metab%2027%3A1212-1221.e3.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cmet.2018.04.010%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cmet.2018.04.010%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Early%20Time-Restricted%20Feeding%20Improves%20Insulin%20Sensitivity%2C%20Blood%20Pressure%2C%20and%20Oxidative%20Stress%20Even%20without%20Weight%20Loss%20in%20Men%20with%20Prediabetes%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Elizabeth%20F.%22%2C%22lastName%22%3A%22Sutton%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Robbie%22%2C%22lastName%22%3A%22Beyl%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kate%20S.%22%2C%22lastName%22%3A%22Early%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22William%20T.%22%2C%22lastName%22%3A%22Cefalu%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Eric%22%2C%22lastName%22%3A%22Ravussin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Courtney%20M.%22%2C%22lastName%22%3A%22Peterson%22%7D%5D%2C%22abstractNote%22%3A%22Intermittent%20fasting%20%28IF%29%20improves%20cardiometabolic%20health%3B%20however%2C%20it%20is%20unknown%20whether%20these%20effects%20are%20due%20solely%20to%20weight%20loss.%20We%20conducted%20the%20first%20supervised%20controlled%20feeding%20trial%20to%20test%20whether%20IF%20has%20benefits%20independent%20of%20weight%20loss%20by%20feeding%20participants%20enough%20food%20to%20maintain%20their%20weight.%20Our%20proof-of-concept%20study%20also%20constitutes%20the%20first%20trial%20of%20early%20time-restricted%20feeding%20%28eTRF%29%2C%20a%20form%20of%20IF%20that%20involves%20eating%20early%20in%20the%20day%20to%20be%20in%20alignment%20with%20circadian%20rhythms%20in%20metabolism.%20Men%20with%20prediabetes%20were%20randomized%20to%20eTRF%20%286-hr%20feeding%20period%2C%20with%20dinner%20before%203%20p.m.%29%20or%20a%20control%20schedule%20%2812-hr%20feeding%20period%29%20for%205%5Cu00a0weeks%20and%20later%20crossed%20over%20to%20the%20other%20schedule.%20eTRF%20improved%20insulin%20sensitivity%2C%20%5Cu03b2%20cell%20responsiveness%2C%20blood%20pressure%2C%20oxidative%20stress%2C%20and%20appetite.%20We%20demonstrate%20for%20the%20first%20time%20in%20humans%20that%20eTRF%20improves%20some%20aspects%20of%20cardiometabolic%20health%20and%20that%20IF%27s%20effects%20are%20not%20solely%20due%20to%20weight%20loss.%22%2C%22date%22%3A%2206%2005%2C%202018%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.cmet.2018.04.010%22%2C%22ISSN%22%3A%221932-7420%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22UIM8YXRX%22%2C%22JFDMJE8P%22%5D%2C%22dateModified%22%3A%222019-06-06T03%3A18%3A51Z%22%7D%7D%2C%7B%22key%22%3A%22XIPWH7MH%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Gabel%20et%20al.%22%2C%22parsedDate%22%3A%222018-06-15%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3EGabel%20K%2C%20Hoddy%20KK%2C%20Haggerty%20N%2C%20et%20al%20%282018%29%20Effects%20of%208-hour%20time%20restricted%20feeding%20on%20body%20weight%20and%20metabolic%20disease%20risk%20factors%20in%20obese%20adults%3A%20A%20pilot%20study.%20Nutr%20Healthy%20Aging%204%3A345%26%23x2013%3B353.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3233%5C%2FNHA-170036%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.3233%5C%2FNHA-170036%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Effects%20of%208-hour%20time%20restricted%20feeding%20on%20body%20weight%20and%20metabolic%20disease%20risk%20factors%20in%20obese%20adults%3A%20A%20pilot%20study%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kelsey%22%2C%22lastName%22%3A%22Gabel%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kristin%20K.%22%2C%22lastName%22%3A%22Hoddy%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Nicole%22%2C%22lastName%22%3A%22Haggerty%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jeehee%22%2C%22lastName%22%3A%22Song%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Cynthia%20M.%22%2C%22lastName%22%3A%22Kroeger%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22John%20F.%22%2C%22lastName%22%3A%22Trepanowski%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Satchidananda%22%2C%22lastName%22%3A%22Panda%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Krista%20A.%22%2C%22lastName%22%3A%22Varady%22%7D%5D%2C%22abstractNote%22%3A%22BACKGROUND%3A%20Time%20restricted%20feeding%20decreases%20energy%20intake%20without%20calorie%20counting%20and%20may%20be%20a%20viable%20option%20for%20weight%20loss.%20However%2C%20the%20effect%20of%20this%20diet%20on%20body%20weight%20in%20obese%20subjects%20has%20never%20been%20examined.%5CnOBJECTIVE%3A%20This%20study%20investigated%20the%20effects%20of%208-h%20time%20restricted%20feeding%20on%20body%20weight%20and%20metabolic%20disease%20risk%20factors%20in%20obese%20adults.%5CnDESIGN%3A%20Obese%20subjects%20%28n%5Cu200a%3D%5Cu200a23%29%20participated%20in%20an%208-h%20time%20restricted%20feeding%20intervention%20%28ad%20libitum%20feeding%20between%2010%3A00%20to%2018%3A00%5Cu200ah%2C%20water%20fasting%20between%2018%3A00%20to%2010%3A00%5Cu200ah%29%20for%2012%20weeks.%20Weight%20loss%20and%20other%20outcomes%20were%20compared%20to%20a%20matched%20historical%20control%20group%20%28n%5Cu200a%3D%5Cu200a23%29.%5CnRESULTS%3A%20Body%20weight%20and%20energy%20intake%20decreased%20in%20the%20time%20restricted%20group%20%28-2.6%25%20%5Cu00b1%200.5%3B%20-341%20%5Cu00b1%2053%5Cu200akcal%5C%2Fd%29%20relative%20to%20controls%20over%2012%20weeks%20%28P%5Cu200a%3C%5Cu200a0.05%29.%20Systolic%20blood%20pressure%20decreased%20in%20the%20time%20restricted%20feeding%20group%20%28-7%20%5Cu00b1%202%5Cu200amm%20Hg%29%20versus%20controls%20%28P%5Cu200a%3C%5Cu200a0.05%29.%20Fat%20mass%2C%20lean%20mass%2C%20visceral%20fat%20mass%2C%20diastolic%20blood%20pressure%2C%20LDL%20cholesterol%2C%20HDL%20cholesterol%2C%20triglycerides%2C%20fasting%20glucose%2C%20fasting%20insulin%2C%20HOMA-IR%2C%20and%20homocysteine%20were%20not%20significantly%20different%20from%20controls%20after%2012%20weeks%20%28no%20group%5Cu00d7time%20interaction%29.%5CnCONCLUSION%3A%20These%20findings%20suggest%20that%208-h%20time%20restricted%20feeding%20produces%20mild%20caloric%20restriction%20and%20weight%20loss%2C%20without%20calorie%20counting.%20It%20may%20also%20offer%20clinical%20benefits%20by%20reducing%20blood%20pressure.%22%2C%22date%22%3A%22Jun%2015%2C%202018%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.3233%5C%2FNHA-170036%22%2C%22ISSN%22%3A%222451-9480%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22UIM8YXRX%22%2C%226BPRM5LB%22%2C%22JQMRJMTS%22%2C%227WXBJPRG%22%2C%22UZR7UXN9%22%5D%2C%22dateModified%22%3A%222019-06-06T03%3A14%3A11Z%22%7D%7D%2C%7B%22key%22%3A%223YQ4G2FK%22%2C%22library%22%3A%7B%22id%22%3A5748374%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Roberts%20et%20al.%22%2C%22parsedDate%22%3A%222017-09-05%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%201.35%3B%20%5C%22%3E%5Cn%20%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%20%20%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%20%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ERoberts%20MN%2C%20Wallace%20MA%2C%20Tomilov%20AA%2C%20et%20al%20%282017%29%20A%20Ketogenic%20Diet%20Extends%20Longevity%20and%20Healthspan%20in%20Adult%20Mice.%20Cell%20Metab%2026%3A539-546.e5.%20%3Ca%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cmet.2017.08.005%27%3Ehttps%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1016%5C%2Fj.cmet.2017.08.005%3C%5C%2Fa%3E%3C%5C%2Fdiv%3E%5Cn%20%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22A%20Ketogenic%20Diet%20Extends%20Longevity%20and%20Healthspan%20in%20Adult%20Mice%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Megan%20N.%22%2C%22lastName%22%3A%22Roberts%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Marita%20A.%22%2C%22lastName%22%3A%22Wallace%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Alexey%20A.%22%2C%22lastName%22%3A%22Tomilov%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Zeyu%22%2C%22lastName%22%3A%22Zhou%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22George%20R.%22%2C%22lastName%22%3A%22Marcotte%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Dianna%22%2C%22lastName%22%3A%22Tran%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gabriella%22%2C%22lastName%22%3A%22Perez%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Elena%22%2C%22lastName%22%3A%22Gutierrez-Casado%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Shinichiro%22%2C%22lastName%22%3A%22Koike%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Trina%20A.%22%2C%22lastName%22%3A%22Knotts%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Denise%20M.%22%2C%22lastName%22%3A%22Imai%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Stephen%20M.%22%2C%22lastName%22%3A%22Griffey%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kyoungmi%22%2C%22lastName%22%3A%22Kim%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Kevork%22%2C%22lastName%22%3A%22Hagopian%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Marissa%20Z.%22%2C%22lastName%22%3A%22McMackin%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Fawaz%20G.%22%2C%22lastName%22%3A%22Haj%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Keith%22%2C%22lastName%22%3A%22Baar%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Gino%20A.%22%2C%22lastName%22%3A%22Cortopassi%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jon%20J.%22%2C%22lastName%22%3A%22Ramsey%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Jose%20Alberto%22%2C%22lastName%22%3A%22Lopez-Dominguez%22%7D%5D%2C%22abstractNote%22%3A%22Calorie%20restriction%2C%20without%20malnutrition%2C%20has%20been%20shown%20to%20increase%20lifespan%20and%20is%20associated%20with%20a%20shift%20away%20from%20glycolysis%20toward%20beta-oxidation.%20The%20objective%20of%20this%20study%20was%20to%20mimic%20this%20metabolic%20shift%20using%20low-carbohydrate%20diets%20and%20to%20determine%20the%20influence%20of%20these%20diets%20on%20longevity%20and%20healthspan%20in%20mice.%20C57BL%5C%2F6%20mice%20were%20assigned%20to%20a%20ketogenic%2C%20low-carbohydrate%2C%20or%20control%20diet%20at%2012%5Cu00a0months%20of%20age%20and%20were%20either%20allowed%20to%20live%20their%5Cu00a0natural%20lifespan%20or%20tested%20for%20physiological%20function%20after%201%20or%2014%5Cu00a0months%20of%20dietary%20intervention.%20The%20ketogenic%20diet%20%28KD%29%20significantly%20increased%20median%20lifespan%20and%20survival%20compared%20to%20controls.%20In%20aged%20mice%2C%20only%20those%20consuming%20a%20KD%20displayed%20preservation%20of%20physiological%20function.%20The%20KD%20increased%20protein%20acetylation%20levels%20and%20regulated%20mTORC1%20signaling%20in%20a%20tissue-dependent%20manner.%20This%20study%20demonstrates%20that%20a%20KD%20extends%20longevity%20and%20healthspan%20in%20mice.%22%2C%22date%22%3A%22Sep%2005%2C%202017%22%2C%22language%22%3A%22eng%22%2C%22DOI%22%3A%2210.1016%5C%2Fj.cmet.2017.08.005%22%2C%22ISSN%22%3A%221932-7420%22%2C%22url%22%3A%22%22%2C%22collections%22%3A%5B%22IB4QNEHC%22%2C%22G7863IM7%22%2C%22BEIYKDRT%22%5D%2C%22dateModified%22%3A%222019-06-03T11%3A39%3A28Z%22%7D%7D%5D%7D
1.
Moro T, Tinsley G, Bianco A, et al (2016) Effects of eight weeks of time-restricted feeding (16/8) on basal metabolism, maximal strength, body composition, inflammation, and cardiovascular risk factors in resistance-trained males. J Transl Med 14:290.
https://doi.org/10.1186/s12967-016-1044-0
1.
Blenkinsopp D The 5-day fast mimicking diet (self experiment). The Quantified Body Podcast.
1.
Woo G mTor Signaling & Cell Growth: Targeting via Ketogenic Diet & Fasting ft. Dr. Keith Baar. HVMN Podcast.
1.
McCay CM, Crowell MF, Maynard LA (1935) The effect of retarded growth upon the length of life span and upon the ultimate body size. J Nutr 10:63–79.
https://doi.org/10.1093/jn/10.1.63
1.
Isley WL, Underwood LE, Clemmons DR (1983) Dietary components that regulate serum somatomedin-C concentrations in humans. J Clin Invest 71:175–182.
https://doi.org/10.1172/jci110757
1.
Sahani MH, Itakura E, Mizushima N (2014) Expression of the autophagy substrate SQSTM1/p62 is restored during prolonged starvation depending on transcriptional upregulation and autophagy-derived amino acids. Autophagy 10:431–441.
https://doi.org/10.4161/auto.27344
1.
Bjørkøy G, Lamark T, Brech A, et al (2005) p62/SQSTM1 forms protein aggregates degraded by autophagy and has a protective effect on huntingtin-induced cell death. J Cell Biol 171:603–614.
https://doi.org/10.1083/jcb.200507002
1.
Seyfried TN, Flores RE, Poff AM, D’Agostino DP (2014) Cancer as a metabolic disease: implications for novel therapeutics. Carcinogenesis 35:515–527.
https://doi.org/10.1093/carcin/bgt480
1.
He C, Bassik MC, Moresi V, et al (2012) Exercise–induced BCL2–regulated autophagy is required for muscle glucose homeostasis. Nature 481:511–515.
https://doi.org/10.1038/nature10758
1.
Clemmons DR, Klibanski A, Underwood LE, et al (1981) Reduction of plasma immunoreactive somatomedin C during fasting in humans. J Clin Endocrinol Metab 53:1247–1250.
https://doi.org/10.1210/jcem-53-6-1247
1.
de Haan G, Nijhof W, Van Zant G (1997) Mouse strain-dependent changes in frequency and proliferation of hematopoietic stem cells during aging: correlation between lifespan and cycling activity. Blood 89:1543–1550
1.
Mitchell SJ, Bernier M, Mattison JA, et al (2019) Daily Fasting Improves Health and Survival in Male Mice Independent of Diet Composition and Calories. Cell Metab 29:221-228.e3.
https://doi.org/10.1016/j.cmet.2018.08.011
1.
Hutchison AT, Regmi P, Manoogian ENC, et al (2019) Time-Restricted Feeding Improves Glucose Tolerance in Men at Risk for Type 2 Diabetes: A Randomized Crossover Trial. Obesity (Silver Spring) 27:724–732.
https://doi.org/10.1002/oby.22449
1.
Wei M, Brandhorst S, Shelehchi M, et al (2017) Fasting-mimicking diet and markers/risk factors for aging, diabetes, cancer, and cardiovascular disease. Science Translational Medicine 9:eaai8700.
https://doi.org/10.1126/scitranslmed.aai8700
1.
Brandhorst S, Choi IY, Wei M, et al (2015) A Periodic Diet that Mimics Fasting Promotes Multi-System Regeneration, Enhanced Cognitive Performance, and Healthspan. Cell Metab 22:86–99.
https://doi.org/10.1016/j.cmet.2015.05.012
1.
Cheng C-W, Adams GB, Perin L, et al (2014) Prolonged fasting reduces IGF-1/PKA to promote hematopoietic-stem-cell-based regeneration and reverse immunosuppression. Cell Stem Cell 14:810–823.
https://doi.org/10.1016/j.stem.2014.04.014
1.
Naito T, Kuma A, Mizushima N (2013) Differential contribution of insulin and amino acids to the mTORC1-autophagy pathway in the liver and muscle. J Biol Chem 288:21074–21081.
https://doi.org/10.1074/jbc.M113.456228
1.
Browning JD, Baxter J, Satapati S, Burgess SC (2012) The effect of short-term fasting on liver and skeletal muscle lipid, glucose, and energy metabolism in healthy women and men. J Lipid Res 53:577–586.
https://doi.org/10.1194/jlr.P020867
1.
Lee C, Safdie FM, Raffaghello L, et al (2010) Reduced levels of IGF-I mediate differential protection of normal and cancer cells in response to fasting and improve chemotherapeutic index. Cancer Res 70:1564–1572.
https://doi.org/10.1158/0008-5472.CAN-09-3228
1.
Carmona-Gutierrez D, Zimmermann A, Kainz K, et al (2019) The flavonoid 4,4’-dimethoxychalcone promotes autophagy-dependent longevity across species. Nat Commun 10:651.
https://doi.org/10.1038/s41467-019-08555-w
1.
Vendelbo MH, Møller AB, Christensen B, et al (2014) Fasting increases human skeletal muscle net phenylalanine release and this is associated with decreased mTOR signaling. PLoS ONE 9:e102031.
https://doi.org/10.1371/journal.pone.0102031
1.
Ezaki J, Matsumoto N, Takeda-Ezaki M, et al (2011) Liver autophagy contributes to the maintenance of blood glucose and amino acid levels. Autophagy 7:727–736.
https://doi.org/10.4161/auto.7.7.15371
1.
Dykstra B, Olthof S, Schreuder J, et al (2011) Clonal analysis reveals multiple functional defects of aged murine hematopoietic stem cells. J Exp Med 208:2691–2703.
https://doi.org/10.1084/jem.20111490
1.
Eisenberg T, Knauer H, Schauer A, et al (2009) Induction of autophagy by spermidine promotes longevity. Nat Cell Biol 11:1305–1314.
https://doi.org/10.1038/ncb1975
1.
Ureta-Vidal A, Ettwiller L, Birney E (2003) Comparative genomics: genome-wide analysis in metazoan eukaryotes. Nat Rev Genet 4:251–262.
https://doi.org/10.1038/nrg1043
1.
Drenick EJ, Alvarez LC, Tamasi GC, Brickman AS (1972) Resistance to symptomatic insulin reactions after fasting. J Clin Invest 51:2757–2762.
https://doi.org/10.1172/JCI107095
1.
LeCheminant JD, Christenson E, Bailey BW, Tucker LA (2013) Restricting night-time eating reduces daily energy intake in healthy young men: a short-term cross-over study. Br J Nutr 110:2108–2113.
https://doi.org/10.1017/S0007114513001359
1.
Sutton EF, Beyl R, Early KS, et al (2018) Early Time-Restricted Feeding Improves Insulin Sensitivity, Blood Pressure, and Oxidative Stress Even without Weight Loss in Men with Prediabetes. Cell Metab 27:1212-1221.e3.
https://doi.org/10.1016/j.cmet.2018.04.010
1.
Gabel K, Hoddy KK, Haggerty N, et al (2018) Effects of 8-hour time restricted feeding on body weight and metabolic disease risk factors in obese adults: A pilot study. Nutr Healthy Aging 4:345–353.
https://doi.org/10.3233/NHA-170036



